Modulation of the activity of cytosolic phospholipase A2alpha (cPLA2alpha) by cellular sphingolipids and inhibition of cPLA2alpha by sphingomyelin
- PMID: 19965591
- PMCID: PMC2842140
- DOI: 10.1194/jlr.M002428
Modulation of the activity of cytosolic phospholipase A2alpha (cPLA2alpha) by cellular sphingolipids and inhibition of cPLA2alpha by sphingomyelin
Abstract
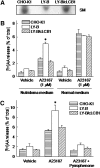
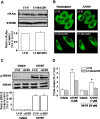
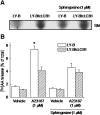
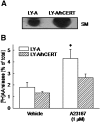
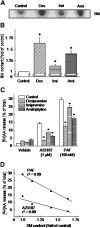
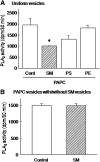
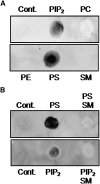
We examined the effect of the cellular sphingolipid level on the release of arachidonic acid (AA) and activity of cytosolic phospholipase A2alpha (cPLA2alpha) using two Chinese hamster ovary (CHO)-K1-derived mutants deficient in sphingolipid synthesis: LY-B cells defective in the LCB1 subunit of serine palmitoyltransferase for de novo synthesis of sphingolipid species, and LY-A cells defective in the ceramide transfer protein CERT for SM synthesis. When LY-B and LY-A cells were cultured in Nutridoma medium and the sphingolipid level was reduced, the release of AA stimulated by the Ca(2+) ionophore A23187 increased 2-fold and 1.7-fold, respectively, compared with that from control cells. The enhancement in LY-B cells was decreased by adding sphingosine and treatment with the cPLA2alpha inhibitor. When CHO cells were treated with an acid sphingomyelinase inhibitor to increase the cellular SM level, the release of AA induced by A23187 or PAF was decreased. In vitro studies were then conducted to test whether SM interacts directly with cPLA2alpha. Phosphatidylcholine vesicles containing SM reduced cPLA2alpha activity. Furthermore, SM disturbed the binding of cPLA2alpha to glycerophospholipids. These results suggest that SM at the biomembrane plays important roles in regulating the cPLA2alpha-dependent release of AA by inhibiting the binding of cPLA2alpha to glycerophospholipids.
Figures
Similar articles
-
Lactosylceramide interacts with and activates cytosolic phospholipase A2α.J Biol Chem. 2013 Aug 9;288(32):23264-72. doi: 10.1074/jbc.M113.491431. Epub 2013 Jun 25. J Biol Chem. 2013. PMID: 23801329 Free PMC article.
-
Sphingomyelin Regulates the Activity of Secretory Phospholipase A2 in the Plasma Membrane.J Cell Biochem. 2015 Sep;116(9):1898-907. doi: 10.1002/jcb.25145. J Cell Biochem. 2015. PMID: 25716287
-
C2-di-ethyl-ceramide-1-phosphate as an inhibitor of group IVA cytosolic phospholipase A2.Eur J Pharmacol. 2012 Dec 15;697(1-3):144-51. doi: 10.1016/j.ejphar.2012.09.041. Epub 2012 Oct 5. Eur J Pharmacol. 2012. PMID: 23043861
-
Role of sphingolipids in arachidonic acid metabolism.J Pharmacol Sci. 2014;124(3):307-12. doi: 10.1254/jphs.13r18cp. Epub 2014 Mar 4. J Pharmacol Sci. 2014. PMID: 24599139 Review.
-
AKT and cytosolic phospholipase A2α form a positive loop in prostate cancer cells.Curr Cancer Drug Targets. 2015;15(9):781-91. doi: 10.2174/1568009615666150706103234. Curr Cancer Drug Targets. 2015. PMID: 26143945 Review.
Cited by
-
Defense mechanisms promoting tolerance to aggressive Phytophthora species in hybrid poplar.Front Plant Sci. 2022 Oct 13;13:1018272. doi: 10.3389/fpls.2022.1018272. eCollection 2022. Front Plant Sci. 2022. PMID: 36325556 Free PMC article.
-
Cytosolic phospholipase A2 protein as a novel therapeutic target for spinal cord injury.Ann Neurol. 2014 May;75(5):644-58. doi: 10.1002/ana.24134. Epub 2014 Apr 2. Ann Neurol. 2014. PMID: 24623140 Free PMC article.
-
Non-Small Cell Lung Cancer Imaging Using a Phospholipase A2 Activatable Fluorophore.Chem Biomed Imaging. 2024 Jun 20;2(7):490-500. doi: 10.1021/cbmi.4c00026. eCollection 2024 Jul 22. Chem Biomed Imaging. 2024. PMID: 39056064 Free PMC article.
-
Lactosylceramide interacts with and activates cytosolic phospholipase A2α.J Biol Chem. 2013 Aug 9;288(32):23264-72. doi: 10.1074/jbc.M113.491431. Epub 2013 Jun 25. J Biol Chem. 2013. PMID: 23801329 Free PMC article.
-
The Involvement of Lactosylceramide in Central Nervous System Inflammation Related to Neurodegenerative Disease.Front Aging Neurosci. 2021 Jul 19;13:691230. doi: 10.3389/fnagi.2021.691230. eCollection 2021. Front Aging Neurosci. 2021. PMID: 34349634 Free PMC article. Review.
References
-
- Uozumi N., Kume K., Nagase T., Nakatani N., Ishii S., Tashiro F., Komagata Y., Maki K., Ikuta K., Ouchi Y., et al. 1997. Role of cytosolic phospholipase A2 in allergic response and parturition. Nature. 390: 618–622 - PubMed
-
- Hirabayashi T., Shimizu T. 2000. Localization and regulation of cytosolic phospholipase A2. Biochim. Biophys. Acta. 1488: 124–138 - PubMed
-
- Hirabayashi T., Murayama T., Shimizu T. 2004. Regulatory mechanism and physiological role of cytosolic phospholipase A2. Biol. Pharm. Bull. 27: 1168–1173 - PubMed
-
- Dennis E. A.1997. The growing phospholipase A2 superfamily of signal transduction enzymes. Trends Biochem. Sci. 22: 1–2 - PubMed
-
- Lin L. L., Wartmann M., Lin A. Y., Knopf J. L., Seth A., Davis R. J. 1993. cPLA2 is phosphorylated and activated by MAP kinase. Cell. 72: 269–278 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

