Nuclear factor of activated T cells regulates osteopontin expression in arterial smooth muscle in response to diabetes-induced hyperglycemia
- PMID: 19965778
- PMCID: PMC2823568
- DOI: 10.1161/ATVBAHA.109.199299
Nuclear factor of activated T cells regulates osteopontin expression in arterial smooth muscle in response to diabetes-induced hyperglycemia
Abstract
Objective: Hyperglycemia is a recognized risk factor for cardiovascular disease in diabetes. Recently, we reported that high glucose activates the Ca(2+)/calcineurin-dependent transcription factor nuclear factor of activated T cells (NFAT) in arteries ex vivo. Here, we sought to determine whether hyperglycemia activates NFAT in vivo and whether this leads to vascular complications.
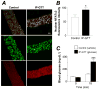
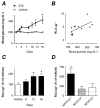
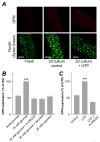
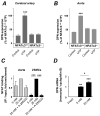
Methods and results: An intraperitoneal glucose-tolerance test in mice increased NFATc3 nuclear accumulation in vascular smooth muscle. Streptozotocin-induced diabetes resulted in increased NFATc3 transcriptional activity in arteries of NFAT-luciferase transgenic mice. Two NFAT-responsive sequences in the osteopontin (OPN) promoter were identified. This proinflammatory cytokine has been shown to exacerbate atherosclerosis and restenosis. Activation of NFAT resulted in increased OPN mRNA and protein in native arteries. Glucose-induced OPN expression was prevented by the ectonucleotidase apyrase, suggesting a mechanism involving the release of extracellular nucleotides. The calcineurin inhibitor cyclosporin A or the novel NFAT blocker A-285222 prevented glucose-induced OPN expression. Furthermore, diabetes resulted in higher OPN expression, which was significantly decreased by in vivo treatment with A-285222 for 4 weeks or prevented in arteries from NFATc3(-/-) mice.
Conclusions: These results identify a glucose-sensitive transcription pathway in vivo, revealing a novel molecular mechanism that may underlie vascular complications of diabetes.
Conflict of interest statement
Figures
Similar articles
-
Inhibition of nuclear factor of activated T-cells (NFAT) suppresses accelerated atherosclerosis in diabetic mice.PLoS One. 2013 Jun 3;8(6):e65020. doi: 10.1371/journal.pone.0065020. Print 2014. PLoS One. 2013. PMID: 23755169 Free PMC article.
-
Nuclear factor of activated T cells is activated in the endothelium of retinal microvessels in diabetic mice.J Diabetes Res. 2015;2015:428473. doi: 10.1155/2015/428473. Epub 2015 Mar 31. J Diabetes Res. 2015. PMID: 25918731 Free PMC article.
-
High glucose activates nuclear factor of activated T cells in native vascular smooth muscle.Arterioscler Thromb Vasc Biol. 2006 Apr;26(4):794-800. doi: 10.1161/01.ATV.0000209513.00765.13. Epub 2006 Feb 9. Arterioscler Thromb Vasc Biol. 2006. PMID: 16469950
-
Endothelin-1 contributes to increased NFATc3 activation by chronic hypoxia in pulmonary arteries.Am J Physiol Cell Physiol. 2011 Aug;301(2):C441-50. doi: 10.1152/ajpcell.00029.2011. Epub 2011 Apr 27. Am J Physiol Cell Physiol. 2011. PMID: 21525433 Free PMC article.
-
Insulin resistance, hyperglycemia, and atherosclerosis.Cell Metab. 2011 Nov 2;14(5):575-85. doi: 10.1016/j.cmet.2011.07.015. Cell Metab. 2011. PMID: 22055501 Free PMC article. Review.
Cited by
-
Relative resistance to Mammalian target of rapamycin inhibition in vascular smooth muscle cells of diabetic donors.Ochsner J. 2013 Spring;13(1):56-60. Ochsner J. 2013. PMID: 23532775 Free PMC article.
-
High glucose enhances store-operated calcium entry by upregulating ORAI/STIM via calcineurin-NFAT signalling.J Mol Med (Berl). 2015 May;93(5):511-21. doi: 10.1007/s00109-014-1234-2. Epub 2014 Dec 5. J Mol Med (Berl). 2015. PMID: 25471481
-
Nuclear factor of activated T cells mediates oxidised LDL-induced calcification of vascular smooth muscle cells.Diabetologia. 2011 Oct;54(10):2690-701. doi: 10.1007/s00125-011-2219-0. Epub 2011 Jun 24. Diabetologia. 2011. PMID: 21701818
-
Inhibition of nuclear factor of activated T-cells (NFAT) suppresses accelerated atherosclerosis in diabetic mice.PLoS One. 2013 Jun 3;8(6):e65020. doi: 10.1371/journal.pone.0065020. Print 2014. PLoS One. 2013. PMID: 23755169 Free PMC article.
-
Determinant of Osteopontin Levels in Microvascular Complications in Patients with Diabetes.Int J Gen Med. 2022 Apr 27;15:4433-4440. doi: 10.2147/IJGM.S354220. eCollection 2022. Int J Gen Med. 2022. PMID: 35509601 Free PMC article.
References
-
- Ohmori R, Momiyama Y, Taniguchi H, Takahashi R, Kusuhara M, Nakamura H, Ohsuzu F. Plasma osteopontin levels are associated with the presence and extent of coronary artery disease. Atherosclerosis. 2003;170:333–337. - PubMed
-
- Kato R, Momiyama Y, Ohmori R, Tanaka N, Taniguchi H, Arakawa K, Kusuhara M, Nakamura H, Ohsuzu F. High plasma levels of osteopontin in patients with restenosis after percutaneous coronary intervention. Arterioscler Thromb Vasc Biol. 2006;26:e1–2. - PubMed
-
- Golledge J, Muller J, Shephard N, Clancy P, Smallwood L, Moran C, Dear AE, Palmer LJ, Norman PE. Association between osteopontin and human abdominal aortic aneurysm. Arterioscler Thromb Vasc Biol. 2007;27:655–660. - PubMed
-
- Scatena M, Liaw L, Giachelli CM. Osteopontin: A Multifunctional Molecule Regulating Chronic Inflammation and Vascular Disease. Arterioscler Thromb Vasc Biol. 2007;27:2302–2309. - PubMed
-
- Strom A, Franzen A, Wangnerud C, Knutsson AK, Heinegard D, Hultgardh-Nilsson A. Altered vascular remodeling in osteopontin-deficient atherosclerotic mice. J Vasc Res. 2004;41:314–322. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

