Difference in desipramine metabolic profile between wild-type and CYP2D6-humanized mice
- PMID: 19995332
- PMCID: PMC2879002
- DOI: 10.2174/187231209790218118
Difference in desipramine metabolic profile between wild-type and CYP2D6-humanized mice
Abstract
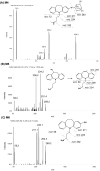
Desipramine (DMI), a CYP2D6 probe, was used as a model drug to test whether CYP2D6-humanized (Tg-CYP2D6) and wild-type control mice could be used as preclinical animal models to identify the effects of CYP2D6 genotype/phenotype on drug metabolic profiles. After the analyses by liquid chromatography coupled with tandem mass spectrometry, DMI biotransformations were compared in Tg-CYP2D6 and wild-type mouse liver microsomes (MLM), and in human CYP2D6 extensive and poor metabolizer liver microsomes. Furthermore, urinary DMI metabolic profiles in Tg-CYP2D6 and wild-type mice were evaluated. Three metabolites, 2-hydroxyl-, 10-hydroxyl, and N-desmethyl-desipramine were identified in the incubations of DMI with both wild-type and Tg-CYP2D6 MLM, as well as in human CYP2D6 extensive metabolizer liver microsomes. Three additional metabolites were found in mouse urine samples, and their chemical structures were elucidated. Although the ratio of individual metabolites produced in Tg-CYP2D6 MLM was closer to that in human CYP2D6 extensive metabolizer liver microsomes, the urinary DMI metabolic profiles did not show much difference between wild-type and Tg-CYP2D6 mice. The results suggest that other mouse enzymes have significant contribution to DMI metabolism.
Figures





Similar articles
-
The Use of In Vitro Data and Physiologically-Based Pharmacokinetic Modeling to Predict Drug Metabolite Exposure: Desipramine Exposure in Cytochrome P4502D6 Extensive and Poor Metabolizers Following Administration of Imipramine.Drug Metab Dispos. 2016 Oct;44(10):1569-78. doi: 10.1124/dmd.116.071639. Epub 2016 Jul 20. Drug Metab Dispos. 2016. PMID: 27440861
-
Venlafaxine: in vitro inhibition of CYP2D6 dependent imipramine and desipramine metabolism; comparative studies with selected SSRIs, and effects on human hepatic CYP3A4, CYP2C9 and CYP1A2.Br J Clin Pharmacol. 1997 Jun;43(6):619-26. doi: 10.1046/j.1365-2125.1997.00591.x. Br J Clin Pharmacol. 1997. PMID: 9205822 Free PMC article.
-
Relationship between plasma desipramine levels, CYP2D6 phenotype and clinical response to desipramine: a prospective study.Eur J Clin Pharmacol. 1997;51(5):395-8. doi: 10.1007/s002280050220. Eur J Clin Pharmacol. 1997. PMID: 9049581
-
Antidepressant drug interactions and the cytochrome P450 system. The role of cytochrome P450 2D6.Clin Pharmacokinet. 1995;29 Suppl 1:10-8; discussion 18-9. doi: 10.2165/00003088-199500291-00004. Clin Pharmacokinet. 1995. PMID: 8846618 Review.
-
Polymorphic cytochrome P450 2D6: humanized mouse model and endogenous substrates.Drug Metab Rev. 2004 May;36(2):243-77. doi: 10.1081/dmr-120034000. Drug Metab Rev. 2004. PMID: 15237854 Review.
Cited by
-
Drug-metabolizing enzyme, transporter, and nuclear receptor genetically modified mouse models.Drug Metab Rev. 2011 Feb;43(1):27-40. doi: 10.3109/03602532.2010.512294. Epub 2010 Sep 21. Drug Metab Rev. 2011. PMID: 20854191 Free PMC article. Review.
-
P450-Humanized and Human Liver Chimeric Mouse Models for Studying Xenobiotic Metabolism and Toxicity.Drug Metab Dispos. 2018 Nov;46(11):1734-1744. doi: 10.1124/dmd.118.083303. Epub 2018 Aug 9. Drug Metab Dispos. 2018. PMID: 30093418 Free PMC article. Review.
-
Humanized transgenic mouse models for drug metabolism and pharmacokinetic research.Curr Drug Metab. 2011 Dec;12(10):997-1006. doi: 10.2174/138920011798062265. Curr Drug Metab. 2011. PMID: 22023319 Free PMC article.
-
Innovative drugs to treat depression: did animal models fail to be predictive or did clinical trials fail to detect effects?Neuropsychopharmacology. 2014 Apr;39(5):1041-51. doi: 10.1038/npp.2013.342. Epub 2013 Dec 18. Neuropsychopharmacology. 2014. PMID: 24345817 Free PMC article. Review.
References
-
- Lin JH. Applications and limitations of interspecies scaling and in vitro extrapolation in pharmacokinetics. Drug Metab Dispos. 1998;26(12):1202–1212. - PubMed
-
- Bogaards JJ, Bertrand M, Jackson P, Oudshoorn MJ, Weaver RJ, van Bladeren PJ, Walther B. Determining the best animal model for human cytochrome P450 activities: a comparison of mouse, rat, rabbit, dog, micropig, monkey and man. Xenobiotica. 2000;30(12):1131–1152. - PubMed
-
- Zanger UM, Raimundo S, Eichelbaum M. Cytochrome P450 2D6: overview and update on pharmacology, genetics, biochemistry. Naunyn Schmiedebergs Arch Pharmacol. 2004;369(1):23–37. - PubMed
-
- Yu AM, Idle JR, Gonzalez FJ. Polymorphic cytochrome P450 2D6: humanized mouse model and endogenous substrates. Drug Metab Rev. 2004;36(2):243–277. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
