Multiple innate signaling pathways cooperate with CD40 to induce potent, CD70-dependent cellular immunity
- PMID: 19995538
- PMCID: PMC2822033
- DOI: 10.1016/j.vaccine.2009.11.071
Multiple innate signaling pathways cooperate with CD40 to induce potent, CD70-dependent cellular immunity
Abstract
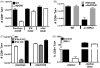
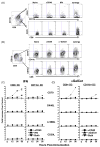
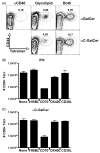
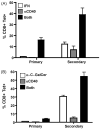
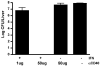
We have previously shown that Toll-like receptor (TLR) agonists cooperate with CD40 to generate CD8 T cell responses exponentially larger than the responses generated with traditional vaccine formulations. We have also shown that combined TLR agonist/anti-CD40 immunization uniquely induces the upregulation of CD70 on antigen bearing dendritic cells (DCs). In contrast, immunization with either a TLR agonist or a CD40 stimulus alone does not significantly increase CD70 expression on DCs. Furthermore, the CD8(+) T cell response generated by combined TLR agonist/anti-CD40 immunization is dependent on the expression of CD70 by DCs, as CD70 blockade following immunization dramatically decreases the CD8 T cell response. Here we show that other innate pathways, independent of the TLRs, can also cooperate with CD40 to induce potent, CD70 dependent, CD8 T cell responses. These innate stimuli include Type I IFN (IFN) and alpha-galactosylceramide (alphaGalCer) or aC-GalCer, glycolipids that are presented by a nonclassical class I MHC molecule, CD1d, and are able to activate NKT cells. Furthermore, this combined IFN/anti-CD40 immunization generates protective memory against bacterial challenge with Listeria monocytogenes. Together these data indicate the importance of assessing CD70 expression on DCs as a marker for the capacity of a given vaccine formulation to potently activate cellular immunity. Our data indicate that optimal induction of CD70 expression requires a coordinated stimulation of both innate (TLR, IFN, alphaGalCer) and adaptive (CD40) signaling pathways.
Copyright (c) 2009 Elsevier Ltd. All rights reserved.
Figures

Similar articles
-
Comparison of OX40 ligand and CD70 in the promotion of CD4+ T cell responses.J Immunol. 2010 Aug 15;185(4):2106-15. doi: 10.4049/jimmunol.1000172. Epub 2010 Jul 16. J Immunol. 2010. PMID: 20639485 Free PMC article.
-
Combined TLR/CD40 stimulation mediates potent cellular immunity by regulating dendritic cell expression of CD70 in vivo.J Immunol. 2007 Feb 1;178(3):1564-72. doi: 10.4049/jimmunol.178.3.1564. J Immunol. 2007. PMID: 17237405
-
CD70 expression by dendritic cells plays a critical role in the immunogenicity of CD40-independent, CD4+ T cell-dependent, licensed CD8+ T cell responses.J Leukoc Biol. 2010 Mar;87(3):477-85. doi: 10.1189/jlb.0809535. Epub 2009 Dec 1. J Leukoc Biol. 2010. PMID: 19952354 Free PMC article.
-
Induction of CD70 on dendritic cells through CD40 or TLR stimulation contributes to the development of CD8+ T cell responses in the absence of CD4+ T cells.J Immunol. 2005 Jan 15;174(2):710-7. doi: 10.4049/jimmunol.174.2.710. J Immunol. 2005. PMID: 15634890
-
Cutting edge: a critical role for CD70 in CD8 T cell priming by CD40-licensed APCs.J Immunol. 2004 Dec 1;173(11):6542-6. doi: 10.4049/jimmunol.173.11.6542. J Immunol. 2004. PMID: 15557144
Cited by
-
Type 1 IFN and PD-L1 Coordinate Lymphatic Endothelial Cell Expansion and Contraction during an Inflammatory Immune Response.J Immunol. 2018 Sep 15;201(6):1735-1747. doi: 10.4049/jimmunol.1800271. Epub 2018 Jul 25. J Immunol. 2018. PMID: 30045970 Free PMC article.
-
Human Anti-CD40 Antibody and Poly IC:LC Adjuvant Combination Induces Potent T Cell Responses in the Lung of Nonhuman Primates.J Immunol. 2015 Aug 1;195(3):1015-24. doi: 10.4049/jimmunol.1500078. Epub 2015 Jun 29. J Immunol. 2015. PMID: 26123354 Free PMC article.
-
Co-administration of α-GalCer analog and TLR4 agonist induces robust CD8(+) T-cell responses to PyCS protein and WT-1 antigen and activates memory-like effector NKT cells.Clin Immunol. 2016 Jul;168:6-15. doi: 10.1016/j.clim.2016.04.014. Epub 2016 Apr 27. Clin Immunol. 2016. PMID: 27132023 Free PMC article.
-
TNF-receptor superfamily agonists as molecular adjuvants for cancer vaccines.Curr Opin Immunol. 2017 Aug;47:70-77. doi: 10.1016/j.coi.2017.07.005. Epub 2017 Jul 24. Curr Opin Immunol. 2017. PMID: 28750279 Free PMC article. Review.
-
Adjuvants for peptide-based cancer vaccines.J Immunother Cancer. 2016 Sep 20;4:56. doi: 10.1186/s40425-016-0160-y. eCollection 2016. J Immunother Cancer. 2016. PMID: 27660710 Free PMC article. Review.
References
-
- Edwards AD, Diebold SS, Slack EM, Tomizawa H, Hemmi H, Kaisho T, Akira S, Reis e Sousa C. Toll-like receptor expression in murine DC subsets: lack of TLR7 expression by CD8 alpha+ DC correlates with unresponsiveness to imidazoquinolines. Eur J Immunol. 2003;33:827–833. - PubMed
-
- Edwards AD, Manickasingham SP, Sporri R, Diebold SS, Schulz O, Sher A, Kaisho T, Akira S, Reis e Sousa C. Microbial recognition via Toll-like receptor-dependent and -independent pathways determines the cytokine response of murine dendritic cell subsets to CD40 triggering. J Immunol. 2002;169:3652–3660. - PubMed
-
- Zhu J, Huang X, Yang Y. Type I IFN signaling on both B and CD4 T cells is required for protective antibody response to adenovirus. J Immunol. 2007;178:3505–3510. - PubMed
-
- Hervas-Stubbs S, Rueda P, Lopez L, Leclerc C. Insect baculoviruses strongly potentiate adaptive immune responses by inducing type I IFN. J Immunol. 2007;178:2361–2369. - PubMed
-
- Fink K, Lang KS, Manjarrez-Orduno N, Junt T, Senn BM, Holdener M, Akira S, Zinkernagel RM, Hengartner H. Early type I interferon-mediated signals on B cells specifically enhance antiviral humoral responses. Eur J Immunol. 2006;36:2094–2105. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

