The rocking bundle: a mechanism for ion-coupled solute flux by symmetrical transporters
- PMID: 19996368
- PMCID: PMC3012352
- DOI: 10.1152/physiol.00030.2009
The rocking bundle: a mechanism for ion-coupled solute flux by symmetrical transporters
Abstract
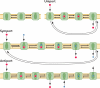
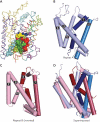
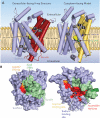
Crystal structures of the bacterial amino acid transporter LeuT have provided the basis for understanding the conformational changes associated with substrate translocation by a multitude of transport proteins with the same fold. Biochemical and modeling studies led to a "rocking bundle" mechanism for LeuT that was validated by subsequent transporter structures. These advances suggest how coupled solute transport might be defined by the internal symmetry of proteins containing inverted structural repeats.
Figures
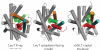
Similar articles
-
Cytoplasmic permeation pathway of neurotransmitter transporters.Biochemistry. 2011 Sep 6;50(35):7462-75. doi: 10.1021/bi200926b. Epub 2011 Aug 10. Biochemistry. 2011. PMID: 21774491 Free PMC article. Review.
-
Atomistic models of ion and solute transport by the sodium-dependent secondary active transporters.Biochim Biophys Acta. 2012 Feb;1818(2):337-47. doi: 10.1016/j.bbamem.2011.10.031. Epub 2011 Nov 29. Biochim Biophys Acta. 2012. PMID: 22138368 Review.
-
Structural elements required for coupling ion and substrate transport in the neurotransmitter transporter homolog LeuT.Proc Natl Acad Sci U S A. 2018 Sep 18;115(38):E8854-E8862. doi: 10.1073/pnas.1716870115. Epub 2018 Sep 4. Proc Natl Acad Sci U S A. 2018. PMID: 30181291 Free PMC article.
-
Uracil/H+ Symport by FurE Refines Aspects of the Rocking-bundle Mechanism of APC-type Transporters.J Mol Biol. 2023 Oct 1;435(19):168226. doi: 10.1016/j.jmb.2023.168226. Epub 2023 Aug 5. J Mol Biol. 2023. PMID: 37544358
-
The structural basis of substrate translocation by the Escherichia coli glycerol-3-phosphate transporter: a member of the major facilitator superfamily.Curr Opin Struct Biol. 2004 Aug;14(4):405-12. doi: 10.1016/j.sbi.2004.06.003. Curr Opin Struct Biol. 2004. PMID: 15313233 Review.
Cited by
-
Fluorescent sensors reporting the activity of ammonium transceptors in live cells.Elife. 2013 Jul 2;2:e00800. doi: 10.7554/eLife.00800. Elife. 2013. PMID: 23840931 Free PMC article.
-
Single-Molecule FRET of Membrane Transport Proteins.Chembiochem. 2021 Sep 2;22(17):2657-2671. doi: 10.1002/cbic.202100106. Epub 2021 May 21. Chembiochem. 2021. PMID: 33945656 Free PMC article. Review.
-
LeuT conformational sampling utilizing accelerated molecular dynamics and principal component analysis.Biophys J. 2012 Jul 3;103(1):L1-3. doi: 10.1016/j.bpj.2012.05.002. Biophys J. 2012. PMID: 22828348 Free PMC article.
-
Model systems for analysis of dopamine transporter function and regulation.Neurochem Int. 2019 Feb;123:13-21. doi: 10.1016/j.neuint.2018.08.015. Epub 2018 Sep 1. Neurochem Int. 2019. PMID: 30179648 Free PMC article. Review.
-
The two Na+ sites in the human serotonin transporter play distinct roles in the ion coupling and electrogenicity of transport.J Biol Chem. 2014 Jan 17;289(3):1825-40. doi: 10.1074/jbc.M113.504654. Epub 2013 Nov 29. J Biol Chem. 2014. PMID: 24293367 Free PMC article.
References
-
- Abramson J, Smirnova I, Kasho V, Verner G, Kaback HR, Iwata S. Structure and mechanism of the lactose permease of Escherichia coli. Science. 2003;301:610–615. - PubMed
-
- Accardi A, Miller C. Secondary active transport mediated by a prokaryotic homologue of ClC Cl– channels. Nature. 2004;427:803–807. - PubMed
-
- Artigas P, Gadsby DC. Ion channel-like properties of the Na+/K+ Pump. Ann NY Acad Sci. 2002;976:31–40. - PubMed
-
- Barrett MP, Walmsley AR, Gould GW. Structure and function of facilitative sugar transporters. Curr Opin Cell Biol. 1999;11:496–502. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

