Bioengineered three-dimensional physiological model of colonic longitudinal smooth muscle in vitro
- PMID: 20001822
- PMCID: PMC2943406
- DOI: 10.1089/ten.TEC.2009.0394
Bioengineered three-dimensional physiological model of colonic longitudinal smooth muscle in vitro
Abstract
Background: The objective of this study was to develop a physiological model of longitudinal smooth muscle tissue from isolated longitudinal smooth muscle cells arranged in the longitudinal axis.
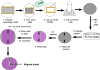
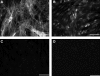
Methods: Longitudinal smooth muscle cells from rabbit sigmoid colon were isolated and expanded in culture. Cells were seeded at high densities onto laminin-coated Sylgard surfaces with defined wavy microtopographies. A highly aligned cell sheet was formed, to which addition of fibrin resulted in delamination.
Results: (1) Acetylcholine (ACh) induced a dose-dependent, rapid, and sustained force generation. (2) Absence of extracellular calcium attenuated the magnitude and sustainability of ACh-induced force by 50% and 60%, respectively. (3) Vasoactive intestinal peptide also attenuated the magnitude and sustainability of ACh-induced force by 40% and 60%, respectively. These data were similar to force generated by longitudinal tissue. (4) Bioengineered constructs also maintained smooth muscle phenotype and calcium-dependence characteristics.
Summary: This is a novel physiologically relevant in vitro three-dimensional model of colonic longitudinal smooth muscle tissue. Bioengineered three-dimensional longitudinal smooth muscle presents the ability to generate force, and respond to contractile agonists and relaxant peptides similar to native longitudinal tissue. This model has potential applications to investigate the underlying pathophysiology of dysfunctional colonic motility. It also presents as a readily implantable band-aid colonic longitudinal muscle tissue.
Figures

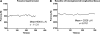
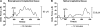
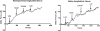
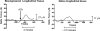
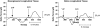
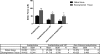
Similar articles
-
Development of a three-dimensional physiological model of the internal anal sphincter bioengineered in vitro from isolated smooth muscle cells.Am J Physiol Gastrointest Liver Physiol. 2005 Aug;289(2):G188-96. doi: 10.1152/ajpgi.00335.2004. Epub 2005 Mar 17. Am J Physiol Gastrointest Liver Physiol. 2005. PMID: 15774939
-
Real-time dynamic movement of caveolin-1 during smooth muscle contraction of human colon and aged rat colon transfected with caveolin-1 cDNA.Am J Physiol Gastrointest Liver Physiol. 2011 Jun;300(6):G1022-32. doi: 10.1152/ajpgi.00301.2010. Epub 2011 Mar 3. Am J Physiol Gastrointest Liver Physiol. 2011. PMID: 21372166 Free PMC article.
-
Mediation of acetylcholine and substance P induced contractions by myosin light chain phosphorylation in feline colonic smooth muscle.Am J Vet Res. 2002 May;63(5):695-702. doi: 10.2460/ajvr.2002.63.695. Am J Vet Res. 2002. PMID: 12013471
-
Chitosan-based scaffolds for the support of smooth muscle constructs in intestinal tissue engineering.Biomaterials. 2012 Jun;33(19):4810-7. doi: 10.1016/j.biomaterials.2012.03.051. Epub 2012 Apr 5. Biomaterials. 2012. PMID: 22483012 Free PMC article.
-
[Capacitative Ca²⁺ entry is involved in ACh-induced distal colon smooth muscle contraction in rats].Sheng Li Xue Bao. 2006 Apr 25;58(2):149-56. Sheng Li Xue Bao. 2006. PMID: 16628362 Chinese.
Cited by
-
BioSphincters to treat Fecal Incontinence in Nonhuman Primates.Sci Rep. 2019 Dec 2;9(1):18096. doi: 10.1038/s41598-019-54440-3. Sci Rep. 2019. PMID: 31792260 Free PMC article.
-
Bioengineering and regeneration of gastrointestinal tissue: where are we now and what comes next?Expert Opin Biol Ther. 2019 Jun;19(6):527-537. doi: 10.1080/14712598.2019.1595579. Epub 2019 Mar 26. Expert Opin Biol Ther. 2019. PMID: 30880502 Free PMC article. Review.
-
Tissue Engineering for Gastrointestinal and Genitourinary Tracts.Int J Mol Sci. 2022 Dec 20;24(1):9. doi: 10.3390/ijms24010009. Int J Mol Sci. 2022. PMID: 36613452 Free PMC article. Review.
-
The influence of extracellular matrix composition on the differentiation of neuronal subtypes in tissue engineered innervated intestinal smooth muscle sheets.Biomaterials. 2014 Aug;35(26):7429-40. doi: 10.1016/j.biomaterials.2014.05.037. Epub 2014 Jun 11. Biomaterials. 2014. PMID: 24929617 Free PMC article.
-
Tissue engineering and regenerative medicine as applied to the gastrointestinal tract.Curr Opin Biotechnol. 2013 Oct;24(5):909-15. doi: 10.1016/j.copbio.2013.03.021. Epub 2013 Apr 10. Curr Opin Biotechnol. 2013. PMID: 23583170 Free PMC article. Review.
References
-
- Sharkey K.A. Kroese A.B.A. Consequences of intestinal inflammation on the enteric nervous system: neuronal activation induced by inflammatory mediators. Anat Rec. 2001;262:79. - PubMed
-
- Straub R.H. Antoniou E. Zeuner M. Gross V. Schölmerich J. Andus T. Association of autonomic nervous hyperreflexia and systemic inflammation in patients with Crohn's disease and ulcerative colitis. J Neuroimmunol. 1997;80:149. - PubMed
-
- Collins S.M. The immunomodulation of enteric neuromuscular function: implications for motility and inflammatory disorders. Gastroenterology. 1996;111:1683. - PubMed
-
- Vermillion D.L. Huizinga J.D. Riddell R.H. Collins S.M. Altered small intestinal smooth muscle function in Crohn's disease. Gastroenterology. 1993;104:1692. - PubMed
-
- Mueller-Klieser W. Three-dimensional cell cultures: from molecular mechanisms to clinical applications. Am J Physiol Cell Physiol. 1997;273:1109. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

