Pharmacokinetic assessment of a five-probe cocktail for CYPs 1A2, 2C9, 2C19, 2D6 and 3A
- PMID: 20002088
- PMCID: PMC2810805
- DOI: 10.1111/j.1365-2125.2009.03548.x
Pharmacokinetic assessment of a five-probe cocktail for CYPs 1A2, 2C9, 2C19, 2D6 and 3A
Abstract
What is already known about this subject: * Numerous cocktails using concurrent administration of several cytochrome P450 (CYP) isoform-selective probe drugs have been reported to investigate drug-drug interactions in vivo. * This approach has several advantages: characterize the inhibitory or induction potential of compounds in development toward the CYP enzymes identified in vitro in an in vivo situation, assess several enzymes in the same trial, and have complete in vivo information about potential CYP-based drug interactions.
What this study adds: * This study describes a new cocktail containing five probe drugs that has never been published. * This cocktail can be used to test the effects of a new chemical entity on multiple CYP isoforms in a single clinical study: CYP1A2 (caffeine), CYP2C9 (warfarin), CYP2C19 (omeprazole), CYP2D6 (metoprolol), and CYP3A (midazolam) and was designed to overcome potential liabilities of other reported cocktails.
Aims: To assess the pharmacokinetics (PK) of selective substrates of CYP1A2 (caffeine), CYP2C9 (S-warfarin), CYP2C19 (omeprazole), CYP2D6 (metoprolol) and CYP3A (midazolam) when administered orally and concurrently as a cocktail relative to the drugs administered alone.
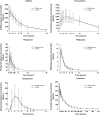
Methods: This was an open-label, single-dose, randomized, six-treatment six-period six-sequence William's design study with a wash-out of 7 or 14 days. Thirty healthy male subjects received 100 mg caffeine, 100 mg metoprolol, 0.03 mg kg(-1) midazolam, 20 mg omeprazole and 10 mg warfarin individually and in combination (cocktail). Poor metabolizers of CYP2C9, 2C19 and 2D6 were excluded. Plasma samples were obtained up to 48 h for caffeine, metoprolol and omeprazole, 12 h for midazolam, 312 h for warfarin and the cocktail. Three different validated liquid chromatography tandem mass spectrometry methods were used. Noncompartmental PK parameters were calculated. Log-transformed C(max), AUC(last) and AUC for each analyte were analysed with a linear mixed effects model with fixed term for treatment, sequence and period, and random term for subject within sequence. Point estimates (90% CI) for treatment ratios (individual/cocktail) were computed for each analyte C(max), AUC(last) and AUC.
Results: There was no PK interaction between the probe drugs when administered in combination as a cocktail, relative to the probes administered alone, as the 90% CI of the PK parameters was within the prespecified bioequivalence limits of 0.80, 1.25.
Conclusion: The lack of interaction between probes indicates that this cocktail could be used to evaluate the potential for multiple drug-drug interactions in vivo.
Figures
References
-
- Chainuvati S, Nafziger AN, Leeder JS, Gaedigk A, Kearns GL, Sellers E, Zhang Y, Kashuba ADM, Rowland E, Bertino JS. Combined phenotypic assessment of CYP1A2, CYP2C9, CYP2C19, CYP2D6, CYP3A, NAT2 and XO with the Cooperstown 5+1 Cocktail. Clin Pharmacol Ther. 2003;74:437–47. - PubMed
-
- Ryu JY, Song IS, Sunwoo YE, Shon JH, Liu KH, Cha IJ, Shin JG. Development of the ‘Inje Cocktail’ for high-throughput evaluation of five human cytochrome P450 isoforms in vivo. Clin Pharmacol Ther. 2007;82:531–40. - PubMed
-
- Tomalik-Scharte D, Jetter A, Kinzig-Schippers M, Skott A, Sörgel F, Klaassen T, Kasel D, Harlfinger S, Doroshyenko O, Frank D, Kirchheiner J, Bräter M, Richter K, Gramatté T, Fuhr U. Effect of propiverine on cytochrome P450 enzymes: a cocktail interaction study in healthy volunteers. Drug Metab Dispos. 2005;33:1859–66. - PubMed
-
- Zhou H, Tong Z, McLeod JF. ‘Cocktail’ approaches and strategies in drug development: valuable tool or flawed science? J Clin Pharmacol. 2004;44:120–34. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical