A changing picture of shigellosis in southern Vietnam: shifting species dominance, antimicrobial susceptibility and clinical presentation
- PMID: 20003464
- PMCID: PMC2803792
- DOI: 10.1186/1471-2334-9-204
A changing picture of shigellosis in southern Vietnam: shifting species dominance, antimicrobial susceptibility and clinical presentation
Abstract
Background: Shigellosis remains considerable public health problem in some developing countries. The nature of Shigellae suggests that they are highly adaptable when placed under selective pressure in a human population. This is demonstrated by variation and fluctuations in serotypes and antimicrobial resistance profile of organisms circulating in differing setting in endemic locations. Antimicrobial resistance in the genus Shigella is a constant threat, with reports of organisms in Asia being resistant to multiple antimicrobials and new generation therapies.
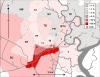
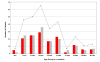
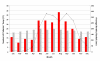
Methods: Here we compare microbiological, clinical and epidemiological data from patients with shigellosis over three different periods in southern Vietnam spanning 14 years.
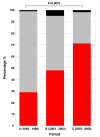
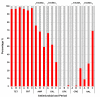
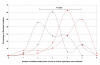
Results: Our data demonstrates a shift in dominant infecting species (S. flexneri to S. sonnei) and resistance profile of the organisms circulating in southern Vietnam. We find that there was no significant variation in the syndromes associated with either S. sonnei or S. flexneri, yet the clinical features of the disease are more severe in later observations.
Conclusions: Our findings show a change in clinical presentation of shigellosis in this setting, as the disease may be now more pronounced, this is concurrent with a change in antimicrobial resistance profile. These data highlight the socio-economic development of southern Vietnam and should guide future vaccine development and deployment strategies.
Trial registration: Current Controlled Trials ISRCTN55945881.
Figures
References
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

