Developmental expression of BK channels in chick cochlear hair cells
- PMID: 20003519
- PMCID: PMC2803478
- DOI: 10.1186/1471-213X-9-67
Developmental expression of BK channels in chick cochlear hair cells
Abstract
Background: Cochlear hair cells are high-frequency sensory receptors. At the onset of hearing, hair cells acquire fast, calcium-activated potassium (BK) currents, turning immature spiking cells into functional receptors. In non-mammalian vertebrates, the number and kinetics of BK channels are varied systematically along the frequency-axis of the cochlea giving rise to an intrinsic electrical tuning mechanism. The processes that control the appearance and heterogeneity of hair cell BK currents remain unclear.
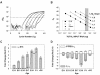
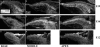
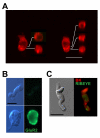
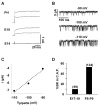
Results: Quantitative PCR results showed a non-monotonic increase in BK alpha subunit expression throughout embryonic development of the chick auditory organ (i.e. basilar papilla). Expression peaked near embryonic day (E) 19 with six times the transcript level of E11 sensory epithelia. The steady increase in gene expression from E11 to E19 could not explain the sudden acquisition of currents at E18-19, implicating post-transcriptional mechanisms. Protein expression also preceded function but progressed in a sequence from diffuse cytoplasmic staining at early ages to punctate membrane-bound clusters at E18. Electrophysiology data confirmed a continued refinement of BK trafficking from E18 to E20, indicating a translocation of BK clusters from supranuclear to subnuclear domains over this critical developmental age.
Conclusions: Gene products encoding BK alpha subunits are detected up to 8 days before the acquisition of anti-BK clusters and functional BK currents. Therefore, post-transcriptional mechanisms seem to play a key role in the delayed emergence of calcium-sensitive currents. We suggest that regulation of translation and trafficking of functional alpha subunits, near voltage-gated calcium channels, leads to functional BK currents at the onset of hearing.
Figures

Similar articles
-
Expression of BK-type calcium-activated potassium channel splice variants during chick cochlear development.J Comp Neurol. 2010 Jul 1;518(13):2554-69. doi: 10.1002/cne.22352. J Comp Neurol. 2010. PMID: 20503427 Free PMC article.
-
BK channels mediate cholinergic inhibition of high frequency cochlear hair cells.PLoS One. 2010 Nov 4;5(11):e13836. doi: 10.1371/journal.pone.0013836. PLoS One. 2010. PMID: 21079807 Free PMC article.
-
Localization and developmental expression of BK channels in mammalian cochlear hair cells.Neuroscience. 2005;130(2):475-84. doi: 10.1016/j.neuroscience.2004.09.038. Neuroscience. 2005. PMID: 15664704
-
BK Channels in the Vertebrate Inner Ear.Int Rev Neurobiol. 2016;128:369-99. doi: 10.1016/bs.irn.2016.03.016. Epub 2016 Apr 20. Int Rev Neurobiol. 2016. PMID: 27238269 Review.
-
Mechanisms of hair cell tuning.Annu Rev Physiol. 1999;61:809-34. doi: 10.1146/annurev.physiol.61.1.809. Annu Rev Physiol. 1999. PMID: 10099711 Review.
Cited by
-
Wnt9a Can Influence Cell Fates and Neural Connectivity across the Radial Axis of the Developing Cochlea.J Neurosci. 2017 Sep 13;37(37):8975-8988. doi: 10.1523/JNEUROSCI.1554-17.2017. Epub 2017 Aug 14. J Neurosci. 2017. PMID: 28821654 Free PMC article.
-
Expression of BK-type calcium-activated potassium channel splice variants during chick cochlear development.J Comp Neurol. 2010 Jul 1;518(13):2554-69. doi: 10.1002/cne.22352. J Comp Neurol. 2010. PMID: 20503427 Free PMC article.
-
Localization and proteomic characterization of cholesterol-rich membrane microdomains in the inner ear.J Proteomics. 2014 May 30;103:178-93. doi: 10.1016/j.jprot.2014.03.037. Epub 2014 Apr 5. J Proteomics. 2014. PMID: 24713161 Free PMC article.
-
Plasticity in ion channel expression underlies variation in hearing during reproductive cycles.Curr Biol. 2013 Apr 22;23(8):678-83. doi: 10.1016/j.cub.2013.03.014. Epub 2013 Apr 4. Curr Biol. 2013. PMID: 23562266 Free PMC article.
-
BDNF profoundly and specifically increases KCNQ4 expression in neurons derived from embryonic stem cells.Stem Cell Res. 2013 Jan;10(1):29-35. doi: 10.1016/j.scr.2012.08.005. Epub 2012 Sep 5. Stem Cell Res. 2013. PMID: 23089626 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

