Developing adenoviral vectors encoding therapeutic genes toxic to host cells: comparing binary and single-inducible vectors expressing truncated E2F-1
- PMID: 20003994
- PMCID: PMC2821996
- DOI: 10.1016/j.virol.2009.11.021
Developing adenoviral vectors encoding therapeutic genes toxic to host cells: comparing binary and single-inducible vectors expressing truncated E2F-1
Erratum in
-
Corrigendum to "Developing adenoviral vectors encoding therapeutic genes toxic to host cells: Comparing binary and single-inducible vectors expressing truncated E2F-1" [Virology 397 (2010) 337-345].Virology. 2025 Feb;603:110355. doi: 10.1016/j.virol.2024.110355. Epub 2024 Dec 16. Virology. 2025. PMID: 39689982 No abstract available.
Abstract
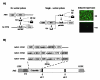
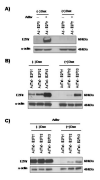
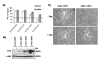
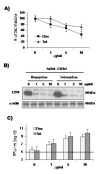
Adenoviral vectors are highly efficient at transferring genes into cells and are broadly used in cancer gene therapy. However, many therapeutic genes are toxic to vector host cells and thus inhibit vector production. The truncated form of E2F-1 (E2Ftr), which lacks the transactivation domain, can significantly induce cancer cell apoptosis, but is also toxic to HEK-293 cells and inhibits adenovirus replication. To overcome this, we have developed binary- and single-vector systems with a modified tetracycline-off inducible promoter to control E2Ftr expression. We compared several vectors and found that the structure of expression cassettes in vectors significantly affects E2Ftr expression. One construct expresses high levels of inducible E2Ftr and efficiently causes apoptotic cancer cell death by activation of caspase-3. The approach developed in this study may be applied in other viral vectors for encoding therapeutic genes that are toxic to their host cells and/or inhibit vector propagation.
Copyright 2009 Elsevier Inc. All rights reserved.
Figures

Similar articles
-
Adenovirus-mediated expression of truncated E2F-1 suppresses tumor growth in vitro and in vivo.Cancer. 2010 Sep 15;116(18):4420-32. doi: 10.1002/cncr.25322. Cancer. 2010. PMID: 20549818 Free PMC article.
-
An adenovirus type 5 (Ad5) amplicon-based packaging cell line for production of high-capacity helper-independent deltaE1-E2-E3-E4 Ad5 vectors.J Virol. 2005 May;79(10):6400-9. doi: 10.1128/JVI.79.10.6400-6409.2005. J Virol. 2005. PMID: 15858023 Free PMC article.
-
A new vector system with inducible E2a cell line for production of higher titer and safer adenoviral vectors.Virology. 2000 Sep 30;275(2):348-57. doi: 10.1006/viro.2000.0515. Virology. 2000. PMID: 10998335
-
Adenovirus as an integrating vector.Curr Gene Ther. 2002 May;2(2):135-44. doi: 10.2174/1566523024605591. Curr Gene Ther. 2002. PMID: 12109211 Review.
-
Adenovirus vectors deleted for genes essential for viral DNA replication.Front Biosci. 2005 May 1;10:1146-55. doi: 10.2741/1607. Front Biosci. 2005. PMID: 15769613 Review.
Cited by
-
Constitutive Gs activation using a single-construct tetracycline-inducible expression system in embryonic stem cells and mice.Stem Cell Res Ther. 2011 Mar 4;2(2):11. doi: 10.1186/scrt52. Stem Cell Res Ther. 2011. PMID: 21375737 Free PMC article.
-
E2F-1 lacking the transcriptional activity domain induces autophagy.Cancer Biol Ther. 2012 Sep;13(11):1091-101. doi: 10.4161/cbt.21143. Epub 2012 Jul 24. Cancer Biol Ther. 2012. PMID: 22825328 Free PMC article.
-
Enhanced cancer cell killing by truncated E2F-1 used in combination with oncolytic adenovirus.Virology. 2012 Nov 25;433(2):538-47. doi: 10.1016/j.virol.2012.09.003. Epub 2012 Sep 27. Virology. 2012. PMID: 23021422 Free PMC article.
-
Adenovirus-mediated expression of truncated E2F-1 suppresses tumor growth in vitro and in vivo.Cancer. 2010 Sep 15;116(18):4420-32. doi: 10.1002/cncr.25322. Cancer. 2010. PMID: 20549818 Free PMC article.
-
E2F-1- and E2Ftr-mediated apoptosis: the role of DREAM and HRK.J Cell Mol Med. 2012 Mar;16(3):605-15. doi: 10.1111/j.1582-4934.2011.01338.x. J Cell Mol Med. 2012. PMID: 21564512 Free PMC article.
References
-
- Bell LA, O’Prey J, Ryan KM. DNA-binding independent cell death from a minimal proapoptotic region of E2F-1. Oncogene. 2006;25(41):5656–63. - PubMed
-
- Dong YB, Phelps AM, Yang HL, Jamshidi-Parsian A, Chen C, Hao H, Gomez-Gutierrez JG, Zhou HS, McMasters KM. Induction of apoptosis signal-regulating Kinase 1 by E2F-1 may not be essential for E2F-1-mediated apoptosis in melanoma cells. Tumour Biol. 2007;28(2):111–22. - PubMed
-
- Dong YB, Yang HL, Elliott MJ, Liu TJ, Stilwell A, Atienza C, Jr., McMasters KM. Adenovirus-mediated E2F-1 gene transfer efficiently induces apoptosis in melanoma cells. Cancer. 1999;86(10):2021–33. - PubMed
-
- Dong YB, Yang HL, Elliott MJ, McMasters KM. Adenovirus-mediated E2F-1 gene transfer sensitizes melanoma cells to apoptosis induced by topoisomerase II inhibitors. Cancer Res. 2002;62(6):1776–83. - PubMed
-
- Elliott MJ, Dong YB, Yang H, McMasters KM. E2F-1 up-regulates c-Myc and p14(ARF) and induces apoptosis in colon cancer cells. Clin Cancer Res. 2001;7(11):3590–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

