The molecular basis of the steep force-calcium relation in heart muscle
- PMID: 20004664
- PMCID: PMC2860225
- DOI: 10.1016/j.yjmcc.2009.11.019
The molecular basis of the steep force-calcium relation in heart muscle
Abstract
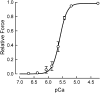
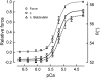
Contraction of heart muscle is regulated by binding of Ca(2+) ions to troponin in the muscle thin filaments, causing a change in filament structure that allows myosin binding and force generation. The steady-state relationship between force and Ca(2+) concentration in demembranated ventricular trabeculae is well described by the Hill equation, with parameters EC(50), the Ca(2+) concentration that gives half the maximum force, and n(H), the Hill coefficient describing the steepness of the Ca(2)(+) dependence. Although each troponin molecule has a single regulatory Ca(2+) site, n(H) is typically around 3, indicating co-operativity in the regulatory mechanism. This review focuses on the molecular basis of this co-operativity, and in particular on the popular hypothesis that force-generating myosin cross-bridges are responsible for the effect. Although cross-bridges can switch on thin filaments at low MgATP concentrations, we argue that the evidence from contracting heart muscle cells shows that this mechanism does not operate in more physiological conditions, and would not play a significant role in the intact heart. Interventions that alter maximum force and EC(50) do not in general produce a significant change in n(H). Complete abolition of force generation by myosin inhibitors does not affect the n(H) values for either Ca(2+) binding to the thin filaments or changes in troponin structure, and both values match that for force generation in the absence of inhibitors. These results provide strong evidence that the co-operative mechanism underlying the high value of n(H) is not due to force-generating cross-bridges but is rather an intrinsic property of the thin filaments.
Copyright (c) 2009 Elsevier Ltd. All rights reserved.
Figures


References
-
- Tobacman L.S. Thin filament-mediated regulation of cardiac contraction. Annu. Rev. Physiol. 1996;58:447–481. - PubMed
-
- Gordon A.M., Homsher E., Regnier M. Regulation of contraction in striated muscle. Physiol. Rev. 2000;80:853–924. - PubMed
-
- Kobayashi T., Solaro R.J. Calcium, thin filaments, and the integrative biology of cardiac contractility. Annu. Rev. Physiol. 2005;67:39–67. - PubMed
-
- Gordon A.M., Regnier M., Homsher E. Skeletal and cardiac muscle contractile activation: tropomyosin “rocks and rolls”. News Physiol. Sci. 2001;16:49–55. - PubMed
-
- Moss R.L., Razumova M., Fitzsimons D.P. Myosin crossbridge activation of cardiac thin filaments: implications for myocardial function in health and disease. Circ. Res. 2004;94:1290–1300. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

