Quantitative expression of VEGF, VEGF-R1, VEGF-R2, and VEGF-R3 in melanoma tissue microarrays
- PMID: 20004943
- PMCID: PMC2824079
- DOI: 10.1016/j.humpath.2009.08.016
Quantitative expression of VEGF, VEGF-R1, VEGF-R2, and VEGF-R3 in melanoma tissue microarrays
Abstract
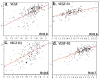
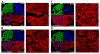
Angiogenesis is required for progression and metastasis of melanoma. Analysis of angiogenic molecules in benign and malignant tissues may allow identification of markers useful for prediction of sensitivity to antiangiogenic agents. We hypothesized that differential expression of vascular endothelial growth factor (VEGF) and its receptors VEGF-R1, VEGF-R2, and VEGF-R3 would be higher in melanomas than nevi and higher in advanced melanoma. Using automated quantitative analysis, we quantified VEGF, -R1, -R2 and -R3 expression in melanoma tissue microarrays composed of 540 nevi and 468 melanoma specimens (198 primaries, 270 metastases). VEGF, VEGF-R1, VEGF-R2, and VEGF-R3 expression was significantly higher in melanomas than nevi by unpaired t tests (P < .0001). VEGF-R2 expression was higher in metastatic specimens (P < .0001), but VEGF-R3 expression was higher in primaries (P < .0001). VEGF was coexpressed with all 3 receptors when assessed by Spearman's rank correlation. VEGF, VEGF-R1, VEGF-R2, and VEGF-R3 expression is higher in melanomas than nevi. Higher expression of VEGF-R2 was found in metastases versus primaries, supporting the idea that selection for an angiogenic phenotype in metastatic melanoma is conferred via up-regulation of VEGF-R2. However, higher expression of VEGF-R3 was seen on primary lesions, potentially implicating this receptor in initiation of lymphatic tumor spread. Clinical trials using antiangiogenic agents in melanoma should include correlative assays of VEGF, VEGF-R1, VEGF-R2, and VEGF-R3 as biomarkers of response to therapy, preferably using quantitative methods such as automated quantitative analysis. Such assessments could assist with evaluation of these molecules as therapeutic targets in melanoma, ultimately facilitating improved selection of patients for treatment.
Copyright 2010 Elsevier Inc. All rights reserved.
Figures


Similar articles
-
Vascular endothelial growth factor receptor expression as a prognostic marker for survival in colorectal cancer.Jpn J Clin Oncol. 2009 Sep;39(9):595-600. doi: 10.1093/jjco/hyp066. Epub 2009 Jun 17. Jpn J Clin Oncol. 2009. PMID: 19535387
-
The role of vascular endothelial growth factors and their receptors in malignant melanomas.Neoplasma. 2008;55(4):273-9. Neoplasma. 2008. PMID: 18505336
-
Explorative investigation of vascular endothelial growth factor receptor expression in primary ovarian cancer and its clinical relevance.Gynecol Oncol. 2014 Jun;133(3):467-72. doi: 10.1016/j.ygyno.2014.03.574. Epub 2014 Apr 5. Gynecol Oncol. 2014. PMID: 24713547
-
CEP-7055: a novel, orally active pan inhibitor of vascular endothelial growth factor receptor tyrosine kinases with potent antiangiogenic activity and antitumor efficacy in preclinical models.Cancer Res. 2003 Sep 15;63(18):5978-91. Cancer Res. 2003. PMID: 14522925
-
The relationship of the angiogenesis regulators VEGF-A, VEGF-R1 and VEGF-R2 to p53 status and prognostic factors in epithelial ovarian carcinoma in FIGO-stages I-II.Int J Oncol. 2016 Mar;48(3):998-1006. doi: 10.3892/ijo.2016.3333. Epub 2016 Jan 12. Int J Oncol. 2016. PMID: 26783205 Free PMC article.
Cited by
-
Endothelin-1 induces the transactivation of vascular endothelial growth factor receptor-3 and modulates cell migration and vasculogenic mimicry in melanoma cells.J Mol Med (Berl). 2013 Mar;91(3):395-405. doi: 10.1007/s00109-012-0956-2. Epub 2012 Sep 11. J Mol Med (Berl). 2013. Retraction in: J Mol Med (Berl). 2024 Nov;102(11):1411. doi: 10.1007/s00109-024-02484-y. PMID: 22965194 Retracted.
-
Clinical Significance of PDCD4 in Melanoma by Subcellular Expression and in Tumor-Associated Immune Cells.Cancers (Basel). 2021 Mar 2;13(5):1049. doi: 10.3390/cancers13051049. Cancers (Basel). 2021. PMID: 33801444 Free PMC article.
-
Vascular endothelial growth factor receptors 1,3 and caveolin-1 are implicated in colorectal cancer aggressiveness and prognosis--correlations with epidermal growth factor receptor, CD44v6, focal adhesion kinase, and c-Met.Tumour Biol. 2013 Aug;34(4):2109-17. doi: 10.1007/s13277-013-0776-1. Epub 2013 Apr 12. Tumour Biol. 2013. PMID: 23580180
-
G-protein-coupled receptor-2-interacting protein-1 is required for endothelial cell directional migration and tumor angiogenesis via cortactin-dependent lamellipodia formation.Arterioscler Thromb Vasc Biol. 2014 Feb;34(2):419-26. doi: 10.1161/ATVBAHA.113.302689. Epub 2013 Nov 21. Arterioscler Thromb Vasc Biol. 2014. PMID: 24265417 Free PMC article.
-
Overexpression of Receptor Tyrosine Kinase EphB4 Triggers Tumor Growth and Hypoxia in A375 Melanoma Xenografts: Insights from Multitracer Small Animal Imaging Experiments.Molecules. 2018 Feb 17;23(2):444. doi: 10.3390/molecules23020444. Molecules. 2018. PMID: 29462967 Free PMC article.
References
-
- Jemal A, Siegel R, Ward E, et al. Cancer Statistics, 2008. CA Cancer J Clin. 2008;58:71–96. - PubMed
-
- Jain RK, Duda DG, Clark JW, Loeffler JS. Lessons from phase III clinical trials on anti-VEGF therapy for cancer. Nat Clin Pract Oncol. 2006;3(1):24–40. - PubMed
-
- Escudier B, Koralewski P, Pluzanska A, et al. A randomized, controlled, double-blind phase III study (AVOREN) of bevacizumab/interferon-a2a vs placebo/interferon- a2a as first-line therapy in metastatic renal cell carcinoma. J Clin Oncol. 2007;25:2s. Abstract.
-
- Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669–76. - PubMed
-
- Einspahr JG, Thomas TL, Saboda K, Nickolof BJ, Warneke J, Curiel-Lewandrowski C, Ranger-Moore J, Duckett L, Bangert J, Fruehauf JP, Alberts DS. Expression of vascular endothelial growth factor in early cutaneous melanocytic lesion progression. Cancer. 2007 Dec 1;110(11):2519–27. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

