Aldehyde dehydrogenase 2 in cardiac protection: a new therapeutic target?
- PMID: 20005475
- PMCID: PMC2856486
- DOI: 10.1016/j.tcm.2009.09.003
Aldehyde dehydrogenase 2 in cardiac protection: a new therapeutic target?
Abstract
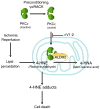
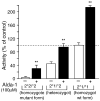
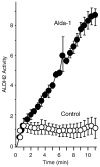
Mitochondrial aldehyde dehydrogenase 2 (ALDH2) is emerging as a key enzyme involved in cytoprotection in the heart. ALDH2 mediates both the detoxification of reactive aldehydes such as acetaldehyde and 4-hydroxy-2-nonenal and the bioactivation of nitroglycerin to nitric oxide. In addition, chronic nitrate treatment results in ALDH2 inhibition and contributes to nitrate tolerance. Our laboratory recently identified ALDH2 to be a key mediator of endogenous cytoprotection. We reported that ALDH2 is phosphorylated and activated by the survival kinase protein kinase C epsilon and found a strong inverse correlation between ALDH2 activity and infarct size. We also identified a small molecule ALDH2 activator which reduces myocardial infarct size induced by ischemia/reperfusion in vivo. In this review, we discuss evidence that ALDH2 is a key mediator of endogenous survival signaling in the heart, suggest possible cardioprotective mechanisms mediated by ALDH2 and discuss potential clinical implications of these findings.
Figures


References
-
- Armstrong S, Downey JM, Ganote CE. Preconditioning of isolated rabbit cardiomyocytes: induction by metabolic stress and blockade by the adenosine antagonist SPT and calphostin C, a protein kinase C inhibitor. Cardiovasc Res. 1994;28:72–7. - PubMed
-
- Baines CP, Kaiser RA, Purcell NH, Blair NS, Osinska H, Hambleton MA, et al. Loss of cyclophilin D reveals a critical role for mitochondrial permeability transition in cell death. Nature. 2005;434:658–62. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

