IFN regulatory factor 5 is required for disease development in the FcgammaRIIB-/-Yaa and FcgammaRIIB-/- mouse models of systemic lupus erythematosus
- PMID: 20007534
- PMCID: PMC2858062
- DOI: 10.4049/jimmunol.0901748
IFN regulatory factor 5 is required for disease development in the FcgammaRIIB-/-Yaa and FcgammaRIIB-/- mouse models of systemic lupus erythematosus
Abstract
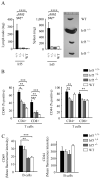
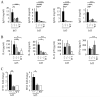
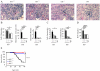
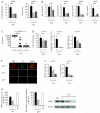
Polymorphisms in the transcription factor IFN regulatory factor 5 (IRF5) are strongly associated in human genetic studies with an increased risk of developing the autoimmune disease systemic lupus erythematosus. However, the biological role of IRF5 in lupus pathogenesis has not previously been tested in an animal model. In this study, we show that IRF5 is absolutely required for disease development in the FcgammaRIIB(-/-)Yaa and FcgammaRIIB(-/-) lupus models. In contrast to IRF5-sufficient FcgammaRIIB(-/-)Yaa mice, IRF5-deficient FcgammaRIIB(-/-)Yaa mice do not develop lupus manifestations and have a phenotype comparable to wild-type mice. Strikingly, full expression of IRF5 is required for the development of autoimmunity, as IRF5 heterozygotes had dramatically reduced disease. One effect of IRF5 is to induce the production of the type I IFN, IFN-alpha, a cytokine implicated in lupus pathogenesis. To address the mechanism by which IRF5 promotes disease, we evaluated FcgammaRIIB(-/-)Yaa mice lacking the type I IFN receptor subunit 1. Unlike the IRF5-deficient and IRF5-heterozygous FcgammaRIIB(-/-)Yaa mice, type I IFN receptor subunit 1-deficient FcgammaRIIB(-/-)Yaa mice maintained a substantial level of residual disease. Furthermore, in FcgammaRIIB(-/-) mice lacking Yaa, IRF5-deficiency also markedly reduced disease manifestations, indicating that the beneficial effects of IRF5 deficiency in FcgammaRIIB(-/-)Yaa mice are not due only to inhibition of the enhanced TLR7 signaling associated with the Yaa mutation. Overall, we demonstrate that IRF5 plays an essential role in lupus pathogenesis in murine models and that this is mediated through pathways beyond that of type I IFN production.
Figures


References
-
- Rhodes B, Vyse TJ. The genetics of SLE: an update in the light of genome-wide association studies. Rheumatology (Oxford) 2008;47:1603–1611. - PubMed
-
- Simard JF, Costenbader KH. What can epidemiology tell us about systemic lupus erythematosus? Int J Clin Pract. 2007;61:1170–1180. - PubMed
-
- Rahman A, Isenberg DA. Systemic lupus erythematosus. N Engl J Med. 2008;358:929–939. - PubMed
-
- Lauwerys BR, Wakeland EK. Genetics of lupus nephritis. Lupus. 2005;14:2–12. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
- AR050942-04/AR/NIAMS NIH HHS/United States
- T32 DK007053/DK/NIDDK NIH HHS/United States
- N01 HV028183/HV/NHLBI NIH HHS/United States
- DK07053/DK/NIDDK NIH HHS/United States
- N01-HV-28183/HV/NHLBI NIH HHS/United States
- R01 AR35230/AR/NIAMS NIH HHS/United States
- P01 AR050256/AR/NIAMS NIH HHS/United States
- AI07290-23/AI/NIAID NIH HHS/United States
- T32 AI007290/AI/NIAID NIH HHS/United States
- R01 AR035230/AR/NIAMS NIH HHS/United States
- T32 AR050942/AR/NIAMS NIH HHS/United States
- N01 HV028183/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

