FLT3-mutant allelic burden and clinical status are predictive of response to FLT3 inhibitors in AML
- PMID: 20007803
- PMCID: PMC2826764
- DOI: 10.1182/blood-2009-09-242859
FLT3-mutant allelic burden and clinical status are predictive of response to FLT3 inhibitors in AML
Abstract
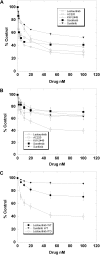
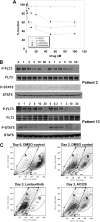
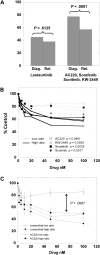
We examined 6 different FMS-like tyrosine kinase-3 (FLT3) inhibitors (lestaurtinib, midostaurin, AC220, KW-2449, sorafenib, and sunitinib) for potency against mutant and wild-type FLT3, as well as for cytotoxic effect against a series of primary blast samples obtained from patients with acute myeloid leukemia (AML) harboring internal tandem duplication (FLT3/ITD) mutations. We found that inhibition of FLT3 autophosphorylation in a FLT3/ITD specimen does not always induce cell death, suggesting that some FLT3/ITD AML may not be addicted to FLT3 signaling. Relapsed samples and samples with a high mutant allelic burden were more likely to be responsive to cytotoxicity from FLT3 inhibition compared with the samples obtained at diagnosis or those with a low mutant allelic burden. These FLT3 inhibitors varied to a considerable degree in their selectivity for FLT3, and this selectivity influenced the cytotoxic effect. These results have important implications for the potential therapeutic use of FLT3 inhibitors in that patients with newly diagnosed FLT3-mutant AML might be less likely to respond clinically to highly selective FLT3 inhibition.
Figures

References
-
- Levis M, Small D. FLT3: ITDoes matter in leukemia. Leukemia. 2003;17(9):1738–1752. - PubMed
-
- Levis M, Small D. FLT3 tyrosine kinase inhibitors. Int J Hematol. 2005;82(2):100–107. - PubMed
-
- Knapper S. FLT3 inhibition in acute myeloid leukaemia. Br J Haematol. 2007;138(6):687–699. - PubMed
-
- Levis M, Smith BD, Beran M, et al. A randomized, open-label study of lestaurtinib (CEP-701), an oral FLT3 inhibitor, administered in sequence with chemotherapy in patients with relapsed AML harboring FLT3 activating mutations: clinical response correlates with successful FLT3 inhibition [abstract]. Blood. 2005;106:121a. Abstract 403.
-
- Cortes J, Foran J, Devetten M, et al. Human pharmacokinetics of AC220, a potent and selective Class III receptor tyrosine kinase inhibitor [abstract]. Blood. 2007;110(11):477a. Abstract 1597.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

