NMDAR-mediated EPSCs are maintained and accelerate in time course during maturation of mouse and rat auditory brainstem in vitro
- PMID: 20008465
- PMCID: PMC2825610
- DOI: 10.1113/jphysiol.2009.184317
NMDAR-mediated EPSCs are maintained and accelerate in time course during maturation of mouse and rat auditory brainstem in vitro
Abstract
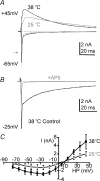
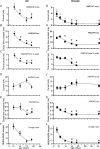
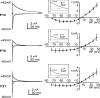
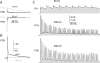
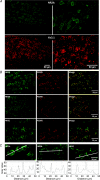
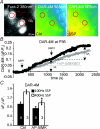
NMDA receptors (NMDARs) mediate a slow EPSC at excitatory glutamatergic synapses throughout the brain. In many areas the magnitude of the NMDAR-mediated EPSC declines with development and is associated with changes in subunit composition, but the mature channel composition is often unknown. We have employed the calyx of Held terminal with its target, the principal neuron of the medial nucleus of the trapezoid body (MNTB), to examine the NMDAR-mediated EPSC during synapse maturation from P10 to P40. Our data show that the calyx has reached a mature state by around P18. The NMDAR-mediated EPSC amplitude (and dominant decay ) fell from around 5 nA (: 40-50 ms) at P10/11 to 0.3-0.5 nA (: 10-15 ms) by P18. The mature NMDAR-EPSC showed no sensitivity to ifenprodil, indicating lack of NR2B subunits, and no block by submicromolar concentrations of zinc, consistent with NR1-1b subunit expression. Additionally, from P11 to P18 there was a reduction in voltage-dependent block and the apparent dissociation constant for [Mg(2+)](o) (K(o)) changed from 7.5 to 14 mm. Quantitative PCR showed that the relative expression of NR2A and NR2C increased, while immunohistochemistry confirmed the presence of NR2A, NR2B and NR2C protein. Although the mature NMDAR-EPSC is small, it is well coupled to NO signalling, as indicated by DAR-4M imaging. We conclude that native mature NMDAR channels at the calyx of Held have a fast time course and reduced block by [Mg(2+)](o), consistent with dominance of NR2C subunits and functional exclusion of NR2B subunits. The pharmacology suggests a single channel type and we postulate that the mature NMDARs consist of heterotrimers of NR1-1b-NR2A-NR2C.
Figures

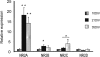
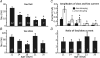
References
-
- Aamodt SM, Constantine-Paton M. The role of neural activity in synaptic development and its implications for adult brain function. Adv Neurol. 1999;79:133–144. - PubMed
-
- Billups B, Wong AC, Forsythe ID. Detecting synaptic connections in the medial nucleus of the trapezoid body using calcium imaging. Pflugers Arch. 2002;444:663–669. - PubMed
-
- Birch PJ, Grossman CJ, Hayes AG. 6,7-Dinitro-quinoxaline-2,3-dion and 6-nitro,7-cyano-quinoxaline-2,3-dion antagonise responses to NMDA in the rat spinal cord via an action at the strychnine-insensitive glycine receptor. Eur J Pharmacol. 1988;156:177–180. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

