Resistance to Trastuzumab in Breast Cancer
- PMID: 20008848
- PMCID: PMC3471537
- DOI: 10.1158/1078-0432.CCR-09-0636
Resistance to Trastuzumab in Breast Cancer
Abstract
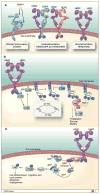
HER2 is a transmembrane oncoprotein encoded by the HER2/neu gene and is overexpressed in approximately 20 to 25% of invasive breast cancers. It can be therapeutically targeted by trastuzumab, a humanized IgG1 kappa light chain monoclonal antibody. Although trastuzumab is currently considered one of the most effective treatments in oncology, a significant number of patients with HER2-overexpressing breast cancer do not benefit from it. Understanding the mechanisms of action and resistance to trastuzumab is therefore crucial for the development of new therapeutic strategies. This review discusses proposed trastuzumab mode of action as well as proposed mechanisms for resistance. Mechanisms for resistance are grouped into four main categories: (1) obstacles preventing trastuzumab binding to HER2; (2) upregulation of HER2 downstream signaling pathways; (3) signaling through alternate pathways; and (4) failure to trigger an immune-mediated mechanism to destroy tumor cells. These potential mechanisms through which trastuzumab resistance may arise have been used as a guide to develop drugs, presently in clinical trials, to overcome resistance. The mechanisms conferring trastuzumab resistance, when completely understood, will provide insight on how best to treat HER2-overexpressing breast cancer. The understanding of each mechanism of resistance is therefore critical for the educated development of strategies to overcome it, as well as for the development of tools that would allow definitive and efficient patient selection for each therapy. (Clin Cancer Res 2009;15(24):7479-91).
Conflict of interest statement
The authors have no potential conflicts of interest to disclose.
Figures





References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

