Early exposure of the pregestational intrauterine and postnatal growth-restricted female offspring to a peroxisome proliferator-activated receptor-{gamma} agonist
- PMID: 20009032
- PMCID: PMC2838528
- DOI: 10.1152/ajpendo.00361.2009
Early exposure of the pregestational intrauterine and postnatal growth-restricted female offspring to a peroxisome proliferator-activated receptor-{gamma} agonist
Abstract
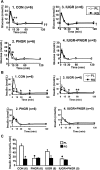
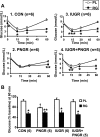
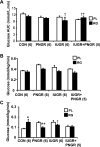
Prenatal nutrient restriction with intrauterine growth restriction (IUGR) alters basal and glucose-stimulated insulin response and hepatic metabolic adaptation. The effect of early intervention with insulin-sensitizing peroxisome proliferator-activated receptor gamma agonists was examined in the metabolically maladapted F(1) pregestational IUGR offspring with a propensity toward pregnancy-induced gestational diabetes. The effect of rosiglitazone maleate [RG; 11 micromol/day from postnatal day (PN) 21 to PN60] vs. placebo (PL) on metabolic adaptations in 2-mo-old F(1) female rats subjected to prenatal (IUGR), postnatal (PNGR), or pre- and postnatal (IUGR + PNGR) nutrient restriction was investigated compared with control (CON). RG vs. PL had no effect on body weight or plasma glucose concentrations but increased subcutaneous white and brown adipose tissue and plasma cholesterol concentrations in all three experimental groups. Glucose tolerance tests with a 1:1 mixture of [2-(2)H(2)]- and [6,6-(2)H(2)]glucose in RG IUGR vs. PL IUGR revealed glucose tolerance with a lower glucose-stimulated insulin release (GSIR) and suppressed endogenous hepatic glucose production (HGP) with no difference in glucose clearance (GC) and recycling (GR). RG PNGR, although similar to PL CON, was hyperglycemic vs. PL PNGR with reduced GR but no difference in the existent low GSIR, HGP, and GC. RG IUGR + PNGR overall was no different from the PL counterpart. Insulin tolerance tests revealed perturbed recovery to baseline from the exaggerated hypoglycemia in RG vs. the PL groups with the only exception being RG PNGR where further worsening of hypoglycemia over PL PNGR was minimal with full recovery to baseline. These observations support that early intervention with RG suppressed HGP in IUGR vs. PL IUGR, without increasing GSIR similar to that seen in CON. Although RG reversed PNGR to the PL CON metabolic state, no such insulin-sensitizing effect was realized in IUGR + PNGR.
Figures
References
-
- Aaboe K, Krarup T, Madsbad S, Holst JJ. GLP-1: physiological effects and potential therapeutic applications. Diabetes Obes Metab 10: 994–1003, 2008 - PubMed
-
- Barker DJ. Adult consequences of fetal growth restriction. Clin Obstet Gynecol 49: 270–283, 2006 - PubMed
-
- Barker DJ, Osmond C, Forsen TJ, Kajantie E, Eriksson JG. Trajectories of growth among children who have coronary events as adults. N Engl J Med 353: 1802–1809, 2005 - PubMed
-
- Bazaes RA, Mericq V. Premature birth and insulin resistance. N Engl J Med 352: 939–940, 2005 - PubMed
-
- Blondeau B, Avril I, Duchene B, Breant B. Endocrine pancreas development is altered in foetuses from rats previously showing intra-uterine growth retardation in response to malnutrition. Diabetologia 45: 394–401, 2002 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

