Chimeric composite skin substitutes for delivery of autologous keratinocytes to promote tissue regeneration
- PMID: 20010085
- PMCID: PMC3015240
- DOI: 10.1097/SLA.0b013e3181c1ab5f
Chimeric composite skin substitutes for delivery of autologous keratinocytes to promote tissue regeneration
Abstract
Objective: We hypothesize that the pathogen-free NIKS human keratinocyte progenitor cell line cultured in a chimeric fashion with patient's primary keratinocytes would produce a fully stratified engineered skin substitute tissue and serve to deliver autologous keratinocytes to a cutaneous wound.
Summary of background data: Chimeric autologous/allogeneic bioengineered skin substitutes offer an innovative regenerative medicine approach for providing wound coverage and restoring cutaneous barrier function while delivering autologous keratinocytes to the wound site. NIKS keratinocytes are an attractive allogeneic cell source for this application.
Methods: Mixed populations of green fluorescent protein (GFP)-labeled NIKS and unlabeled primary keratinocytes were used to model the allogeneic and autologous components in chimeric monolayer and organotypic cultures.
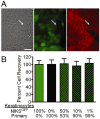
Results: In monolayer coculture, GFP-labeled NIKS had no effect on the growth rate of primary keratinocytes and cell-cell junction formation between labeled and unlabeled keratinocytes was observed. In organotypic culture employing dermal and epidermal compartments, chimeric composite skin substitutes generated using up to 90% GFP-labeled NIKS exhibited normal tissue architecture and possessed substantial regions attributable to the primary keratinocytes. Tissues expressed proteins essential for the structure and function of a contiguous, fully-stratified squamous epithelia and exhibited barrier function similar to that of native skin. Furthermore, chimeric human skin substitutes stably engrafted in an in vivo mouse model, with long-term retention of primary keratinocytes but loss of the GFP-labeled NIKS population by 28 days after surgical application.
Conclusions: This study provides proof of concept for the use of NIKS keratinocytes as an allogeneic cell source for the formation of bioengineered chimeric skin substitute tissues, providing immediate formal wound coverage while simultaneously supplying autologous cells for tissue regeneration.
Figures

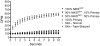




Similar articles
-
Chimeric Human Skin Substitute Tissue: A Novel Treatment Option for the Delivery of Autologous Keratinocytes.Adv Wound Care (New Rochelle). 2012 Apr;1(2):57-62. doi: 10.1089/wound.2011.0340. Adv Wound Care (New Rochelle). 2012. PMID: 24527281 Free PMC article.
-
Chimeric autologous/allogeneic constructs for skin regeneration.Mil Med. 2014 Aug;179(8 Suppl):71-8. doi: 10.7205/MILMED-D-13-00480. Mil Med. 2014. PMID: 25102552
-
StrataGraft skin substitute is well-tolerated and is not acutely immunogenic in patients with traumatic wounds: results from a prospective, randomized, controlled dose escalation trial.Ann Surg. 2011 Apr;253(4):672-83. doi: 10.1097/SLA.0b013e318210f3bd. Ann Surg. 2011. PMID: 21475006 Free PMC article. Clinical Trial.
-
Tissue-engineered skin substitutes in regenerative medicine.Curr Opin Biotechnol. 2009 Oct;20(5):563-7. doi: 10.1016/j.copbio.2009.08.008. Epub 2009 Sep 24. Curr Opin Biotechnol. 2009. PMID: 19782559 Review.
-
Skin tissue engineering.Clin Plast Surg. 2003 Oct;30(4):573-9. doi: 10.1016/s0094-1298(03)00075-0. Clin Plast Surg. 2003. PMID: 14621305 Review.
Cited by
-
Visualization of morphological and molecular features associated with chronic ischemia in bioengineered human skin.Microsc Microanal. 2010 Apr;16(2):117-31. doi: 10.1017/S1431927610000103. Epub 2010 Mar 4. Microsc Microanal. 2010. PMID: 20199713 Free PMC article.
-
Chimeric Human Skin Substitute Tissue: A Novel Treatment Option for the Delivery of Autologous Keratinocytes.Adv Wound Care (New Rochelle). 2012 Apr;1(2):57-62. doi: 10.1089/wound.2011.0340. Adv Wound Care (New Rochelle). 2012. PMID: 24527281 Free PMC article.
-
Nonviral human beta defensin-3 expression in a bioengineered human skin tissue: a therapeutic alternative for infected wounds.Wound Repair Regen. 2012 May-Jun;20(3):414-24. doi: 10.1111/j.1524-475X.2012.00786.x. Wound Repair Regen. 2012. PMID: 22564233 Free PMC article.
-
Clinical Evaluation of NIKS-Based Bioengineered Skin Substitute Tissue in Complex Skin Defects: Phase I/IIa Clinical Trial Results.Adv Wound Care (New Rochelle). 2012 Apr;1(2):95-103. doi: 10.1089/wound.2011.0343. Adv Wound Care (New Rochelle). 2012. PMID: 24527287 Free PMC article.
-
Vascularization of the dermal support enhances wound re-epithelialization by in situ delivery of epidermal keratinocytes.Tissue Eng Part A. 2011 Mar;17(5-6):665-75. doi: 10.1089/ten.TEA.2010.0125. Epub 2010 Dec 18. Tissue Eng Part A. 2011. PMID: 20929281 Free PMC article.
References
-
- Atiyeh BS, Costagliola M. Cultured epithelial autograft (CEA) in burn treatment: three decades later. Burns. 2007;33(4):405–13. - PubMed
-
- Nemes Z, Steinert PM. Bricks and mortar of the epidermal barrier. Exp Mol Med. 1999;31(1):5–19. - PubMed
-
- Cairns BA, deSerres S, Peterson HD, Meyer AA. Skin replacements. The biotechnological quest for optimal wound closure. Arch Surg. 1993;128(11):1246–52. - PubMed
-
- Suzuki T, Ui K, Shioya N, Ihara S. Mixed cultures comprising syngeneic and allogeneic mouse keratinocytes as a graftable skin substitute. Transplantation. 1995;59(9):1236–41. - PubMed
-
- Rouabhia M. Permanent skin replacement using chimeric epithelial cultured sheets comprising xenogeneic and syngeneic keratinocytes. Transplantation. 1996;61(9):1290–300. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

