The protective effects of alpha-ketoacids against oxidative stress on rat spermatozoa in vitro
- PMID: 20010848
- PMCID: PMC3739092
- DOI: 10.1038/aja.2009.78
The protective effects of alpha-ketoacids against oxidative stress on rat spermatozoa in vitro
Abstract
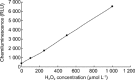
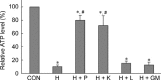
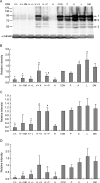
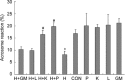
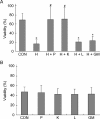
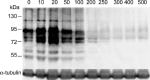
The aim of this study was to determine the effects of antioxidants, including alpha-ketoacids (alpha-ketoglutarate and pyruvate), lactate and glutamate/malate combination, against oxidative stress on rat spermatozoa. Our results showed that H(2)O(2) (250 micromol L(-1))-induced damages, such as impaired motility, adenosine triphosphate (ATP) depletion, inhibition of sperm protein phosphorylation, reduced acrosome reaction and decreased viability, could be significantly prevented by incubation of the spermatozoa with alpha-ketoglutarate (4 mmol L(-1)) or pyruvate (4 mmol L(-1)). Without exogenous H(2)O(2) in the medium, the addition of pyruvate (4 mmol L(-1)) significantly increased the superoxide anion (O(2)(-).) level in sperm suspension (P < or = 0.01), whereas the addition of alpha-ketoglutarate (4 mmol L(-1)) and lactate (4 mmol L(-1)) significantly enhanced tyrosine-phosphorylated proteins with the size of 95 kDa (P < or = 0.04). At the same time, alpha-ketoglutarate, pyruvate, lactate, glutamate and malate supplemented in media can be used as important energy sources and supply ATP for sperm motility. In conclusion, the present results show that alpha-ketoacids could be effective antioxidants for protecting rat spermatozoa from H(2)O(2) attack and could be effective components to improve the antioxidant capacity of Biggers, Whitten and Whittingham media.
Figures



Similar articles
-
Lactate and Pyruvate Are Major Sources of Energy for Stallion Sperm with Dose Effects on Mitochondrial Function, Motility, and ROS Production.Biol Reprod. 2016 Aug;95(2):34. doi: 10.1095/biolreprod.116.140707. Epub 2016 Jun 22. Biol Reprod. 2016. PMID: 27335066
-
Correlation between tyrosine phosphorylation intensity of a 107 kDa protein band and A23187-induced acrosome reaction in human spermatozoa.Andrologia. 2004 Dec;36(6):370-7. doi: 10.1111/j.1439-0272.2004.00634.x. Andrologia. 2004. PMID: 15541053
-
L-carnitine and pyruvate are prosurvival factors during the storage of stallion spermatozoa at room temperature.Biol Reprod. 2015 Oct;93(4):104. doi: 10.1095/biolreprod.115.131326. Epub 2015 Aug 26. Biol Reprod. 2015. PMID: 26316064
-
Peroxiredoxins prevent oxidative stress during human sperm capacitation.Mol Hum Reprod. 2017 Feb 10;23(2):106-115. doi: 10.1093/molehr/gaw081. Mol Hum Reprod. 2017. PMID: 28025393 Free PMC article.
-
Taurine effects on Bisphenol A-induced oxidative stress in the mouse testicular mitochondria and sperm motility.JBRA Assist Reprod. 2020 Oct 6;24(4):428-435. doi: 10.5935/1518-0557.20200017. JBRA Assist Reprod. 2020. PMID: 32550655 Free PMC article. Review.
Cited by
-
Tamoxifen is a potent antioxidant modulator for sperm quality in patients with idiopathic oligoasthenospermia.Int Urol Nephrol. 2015 Sep;47(9):1463-9. doi: 10.1007/s11255-015-1065-2. Epub 2015 Jul 28. Int Urol Nephrol. 2015. PMID: 26216675 Clinical Trial.
-
Oxidative testicular injury: effect of L-leucine on redox, cholinergic and purinergic dysfunctions, and dysregulated metabolic pathways.Amino Acids. 2021 Mar;53(3):359-380. doi: 10.1007/s00726-021-02954-4. Epub 2021 Feb 14. Amino Acids. 2021. PMID: 33586041
-
A scoping review regarding reproductive capacity modulation based on alpha-ketoglutarate supplementation.Reproduction. 2024 Oct 7;168(5):e240137. doi: 10.1530/REP-24-0137. Print 2024 Nov 1. Reproduction. 2024. PMID: 39189990 Free PMC article.
-
Low Protein Diets Supplemented With Alpha-Ketoglutarate Enhance the Growth Performance, Immune Response, and Intestinal Health in Common Carp (Cyprinus carpio).Front Immunol. 2022 Jun 2;13:915657. doi: 10.3389/fimmu.2022.915657. eCollection 2022. Front Immunol. 2022. PMID: 35720284 Free PMC article.
-
Suppressive effect of CORM-2 on LPS-induced platelet activation by glycoprotein mediated HS1 phosphorylation interference.PLoS One. 2013 Dec 20;8(12):e83112. doi: 10.1371/journal.pone.0083112. eCollection 2013. PLoS One. 2013. PMID: 24376647 Free PMC article.
References
-
- Aitken RJ.Sperm function tests and fertility Int J Androl 20062969–75.discussion 105–8. - PubMed
-
- Aitken RJ, Baker MA. Oxidative stress and male reproductive biology. Reprod Fertil Dev. 2004;16:581–8. - PubMed
-
- Tremellen K. Oxidative stress and male infertility—a clinical perspective. Hum Reprod Update. 2008;14:243–58. - PubMed
-
- Rivlin J, Mendel J, Rubinstein S, Etkovitz N, Breitbart H. Role of hydrogen peroxide in sperm capacitation and acrosome reaction. Biol Reprod. 2004;70:518–22. - PubMed
-
- Zalata AA, Ahmed AH, Allamaneni SS, Comhaire FH, Agarwal A. Relationship between acrosin activity of human spermatozoa and oxidative stress. Asian J Androl. 2004;6:313–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

