An alpha-galactosylceramide C20:2 N-acyl variant enhances anti-inflammatory and regulatory T cell-independent responses that prevent type 1 diabetes
- PMID: 20015094
- PMCID: PMC2857941
- DOI: 10.1111/j.1365-2249.2009.04074.x
An alpha-galactosylceramide C20:2 N-acyl variant enhances anti-inflammatory and regulatory T cell-independent responses that prevent type 1 diabetes
Abstract
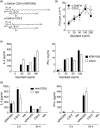
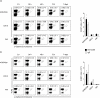
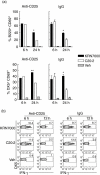
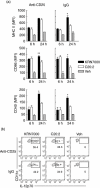
Protection from type 1 diabetes (T1D), a T helper type 1 (Th1)-mediated disease, is achievable in non-obese diabetic (NOD) mice by treatment with alpha-galactosylceramide (alpha-GalCer) glycolipids that stimulate CD1d-restricted invariant natural killer T (iNK T) cells. While we have reported previously that the C20:2 N-acyl variant of alpha-GalCer elicits a Th2-biased cytokine response and protects NOD mice from T1D more effectively than a form of alpha-GalCer that induces mixed Th1 and Th2 responses, it remained to determine whether this protection is accompanied by heightened anti-inflammatory responses. We show that treatment of NOD mice with C20:2 diminished the activation of 'inflammatory' interleukin (IL)-12 producing CD11c(high)CD8+ myeloid dendritic cells (mDCs) and augmented the function of 'tolerogenic' DCs more effectively than treatment with the prototypical iNKT cell activator KRN7000 (alpha-GalCer C26:0) that induces Th1- and Th2-type responses. These findings correlate with a reduced capacity of C20:2 to sustain the early transactivation of T, B and NK cells. They may also explain our observation that C20:2 activated iNK T cells depend less than KRN7000 activated iNK T cells upon regulation by regulatory T cells for cytokine secretion and protection from T1D. The enhanced anti-inflammatory properties of C20:2 relative to KRN7000 suggest that C20:2 should be evaluated further as a drug to induce iNK T cell-mediated protection from T1D in humans.
Figures



Similar articles
-
Stimulation of a shorter duration in the state of anergy by an invariant natural killer T cell agonist enhances its efficiency of protection from type 1 diabetes.Clin Exp Immunol. 2011 Apr;164(1):26-41. doi: 10.1111/j.1365-2249.2011.04323.x. Epub 2011 Mar 1. Clin Exp Immunol. 2011. PMID: 21361909 Free PMC article.
-
Structure-guided design of an invariant natural killer T cell agonist for optimum protection from type 1 diabetes in non-obese diabetic mice.Clin Exp Immunol. 2011 Oct;166(1):121-33. doi: 10.1111/j.1365-2249.2011.04454.x. Clin Exp Immunol. 2011. PMID: 21910729 Free PMC article.
-
Activation of natural killer T cells by α-carba-GalCer (RCAI-56), a novel synthetic glycolipid ligand, suppresses murine collagen-induced arthritis.Clin Exp Immunol. 2011 May;164(2):236-47. doi: 10.1111/j.1365-2249.2011.04369.x. Epub 2011 Mar 10. Clin Exp Immunol. 2011. PMID: 21391989 Free PMC article.
-
Switching invariant natural killer T (iNKT) cell response from anticancerous to anti-inflammatory effect: molecular bases.J Med Chem. 2014 Jul 10;57(13):5489-508. doi: 10.1021/jm4010863. Epub 2014 Feb 12. J Med Chem. 2014. PMID: 24428717 Review.
-
Tailored design of NKT-stimulatory glycolipids for polarization of immune responses.J Biomed Sci. 2017 Mar 23;24(1):22. doi: 10.1186/s12929-017-0325-0. J Biomed Sci. 2017. PMID: 28335781 Free PMC article. Review.
Cited by
-
Design, synthesis, and functional activity of labeled CD1d glycolipid agonists.Bioconjug Chem. 2013 Apr 17;24(4):586-94. doi: 10.1021/bc300556e. Epub 2013 Mar 19. Bioconjug Chem. 2013. PMID: 23458425 Free PMC article.
-
The immunosuppressive role of adenosine A2A receptors in ischemia reperfusion injury and islet transplantation.Curr Diabetes Rev. 2012 Nov;8(6):419-33. doi: 10.2174/157339912803529878. Curr Diabetes Rev. 2012. PMID: 22934547 Free PMC article.
-
Can invariant Natural Killer T cells drive B cell fate? a look at the humoral response.Front Immunol. 2025 Feb 18;16:1505883. doi: 10.3389/fimmu.2025.1505883. eCollection 2025. Front Immunol. 2025. PMID: 40040714 Free PMC article. Review.
-
T helper type 2-polarized invariant natural killer T cells reduce disease severity in acute intra-abdominal sepsis.Clin Exp Immunol. 2014 Nov;178(2):292-309. doi: 10.1111/cei.12404. Clin Exp Immunol. 2014. PMID: 24965554 Free PMC article.
-
Therapeutic manipulation of natural killer (NK) T cells in autoimmunity: are we close to reality?Clin Exp Immunol. 2013 Jan;171(1):8-19. doi: 10.1111/j.1365-2249.2012.04625.x. Clin Exp Immunol. 2013. PMID: 23199318 Free PMC article. Review.
References
-
- Bendelac A, Savage PB, Teyton L. The biology of NKT cells. Annu Rev Immunol. 2007;25:297–336. - PubMed
-
- Kronenberg M. Toward an understanding of NKT cell biology: progress and paradoxes. Annu Rev Immunol. 2005;23:877–900. - PubMed
-
- Van Kaer L. Alpha-galactosylceramide therapy for autoimmune diseases: prospects and obstacles. Nat Rev Immunol. 2005;5:31–42. - PubMed
-
- Godfrey DI, MacDonald HR, Kronenberg M, Smyth MJ, Van Kaer L. NKT cells: what's in a name? Nat Rev Immunol. 2004;4:231–7. - PubMed
-
- Anderson MS, Bluestone JA. The NOD mouse: a model of immune dysregulation. Annu Rev Immunol. 2005;23:447–85. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

