Unique activation status of peripheral blood mononuclear cells at acute phase of Kawasaki disease
- PMID: 20015095
- PMCID: PMC2857948
- DOI: 10.1111/j.1365-2249.2009.04073.x
Unique activation status of peripheral blood mononuclear cells at acute phase of Kawasaki disease
Abstract
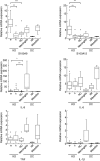
Although Kawasaki disease (KD) is characterized by a marked activation of the immune system with elevations of serum proinflammatory cytokines and chemokines at acute phase, the major sources for these chemical mediators remain controversial. We analysed the activation status of peripheral blood mononuclear cells (PBMCs) by flow cytometry, DNA microarray and quantitative reverse transcription-polymerase chain reaction. The proportions of CD69+ cells in both natural killer cells and gammadeltaT cells at acute-phase KD were significantly higher than those at convalescent-phase KD. Microarray analysis revealed that five genes such as NAIP, IPAF, S100A9, FCGR1A and GCA up-regulated in acute-phase KD and the pathways involved in acute phase KD were related closely to the innate immune system. The relative expression levels of damage-associated molecular pattern molecule (DAMP) (S100A9 and S100A12) genes in PBMCs at acute-phase KD were significantly higher than those at convalescent-phase KD, while those of TNFA, IL1B and IL6 genes were not significantly different between KD patients and healthy controls. Intracellular production of tumour necrosis factor-alpha, interleukin-10 and interferon-gamma in PBMCs was not observed in KD patients. The present data have indicated that PBMCs showed a unique activation status with high expression of DAMP genes but low expression of proinflammatory cytokine genes, and that the innate immune system appears to play a role in the pathogenesis and pathophysiology of KD.
Figures


Similar articles
-
Aberrantly decreased levels of NKG2D expression in children with kawasaki disease.Scand J Immunol. 2013 May;77(5):389-97. doi: 10.1111/sji.12022. Scand J Immunol. 2013. PMID: 23298273
-
Adrenomedullin is highly expressed in blood monocytes associated with acute Kawasaki disease: a microarray gene expression study.Pediatr Res. 2005 Jan;57(1):49-55. doi: 10.1203/01.PDR.0000147745.52711.DD. Epub 2004 Nov 5. Pediatr Res. 2005. PMID: 15531734
-
[Changes and significance for regulatory factors for signal pathways of Toll-like receptors in immunological pathogenesis of Kawasaki disease].Zhonghua Er Ke Za Zhi. 2008 Jan;46(1):49-54. Zhonghua Er Ke Za Zhi. 2008. PMID: 18353240 Chinese.
-
Immunological profile of peripheral blood lymphocytes and monocytes/macrophages in Kawasaki disease.Clin Exp Immunol. 2005 Sep;141(3):381-7. doi: 10.1111/j.1365-2249.2005.02821.x. Clin Exp Immunol. 2005. PMID: 16045726 Free PMC article. Review.
-
[Regulation of proinflammatory cytokine cascade in Kawasaki disease].Nihon Rinsho. 2008 Feb;66(2):258-64. Nihon Rinsho. 2008. PMID: 18260323 Review. Japanese.
Cited by
-
A non-invasive diagnosis of histiocytic necrotizing lymphadenitis by means of gene expression profile analysis of peripheral blood mononuclear cells.J Clin Immunol. 2013 Jul;33(5):1018-26. doi: 10.1007/s10875-013-9897-y. Epub 2013 Apr 23. J Clin Immunol. 2013. PMID: 23609111
-
Suppressed plasmablast responses in febrile infants, including children with Kawasaki disease.PLoS One. 2018 Mar 26;13(3):e0193539. doi: 10.1371/journal.pone.0193539. eCollection 2018. PLoS One. 2018. PMID: 29579044 Free PMC article.
-
Kawasaki disease-specific molecules in the sera are linked to microbe-associated molecular patterns in the biofilms.PLoS One. 2014 Nov 20;9(11):e113054. doi: 10.1371/journal.pone.0113054. eCollection 2014. PLoS One. 2014. PMID: 25411968 Free PMC article.
-
Interleukin-33/ST2 Axis as Potential Biomarker and Therapeutic Target in Kawasaki Disease.Inflammation. 2023 Feb;46(1):480-490. doi: 10.1007/s10753-022-01753-7. Epub 2022 Oct 8. Inflammation. 2023. PMID: 36208354
-
Intravenous Immunoglobulin Therapy Restores the Quantity and Phenotype of Circulating Dendritic Cells and CD4+ T Cells in Children With Acute Kawasaki Disease.Front Immunol. 2022 Feb 10;13:802690. doi: 10.3389/fimmu.2022.802690. eCollection 2022. Front Immunol. 2022. PMID: 35222381 Free PMC article.
References
-
- Burns JC, Glode MP. Kawasaki syndrome. Lancet. 2004;364:533–44. - PubMed
-
- Ichiyama T, Yoshitomi T, Nishikawa M, et al. NF-kappaB activation in peripheral blood monocytes/macrophages and T cells during acute Kawasaki disease. Clin Immunol. 2001;99:373–7. - PubMed
-
- Abe J, Jibiki T, Noma S, Nakajima T, Saito H, Terai M. Gene expression profiling of the effect of high-dose intravenous Ig in patients with Kawasaki disease. J Immunol. 2005;174:5837–45. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

