Myelin protein zero is naturally processed in the B cells of monoclonal gammopathy of undetermined significance of immunoglobulin M isotype: aberrant triggering of a patient's T cells
- PMID: 20015874
- PMCID: PMC2857193
- DOI: 10.3324/haematol.2009.015123
Myelin protein zero is naturally processed in the B cells of monoclonal gammopathy of undetermined significance of immunoglobulin M isotype: aberrant triggering of a patient's T cells
Abstract
Background: Monoclonal gammopathy of undetermined significance of immunoglobulin M isotype is a condition with clonally expanded B cells, recently suggested to have an infectious origin. This monoclonal gammopathy is frequently associated with polyneuropathy and antibodies against myelin protein zero, whereas the role of the T cells remains largely unknown. We analyzed protein zero-specific B cells, as antigen-presenting cells, and their capacity to activate T helper cells.
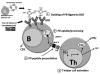
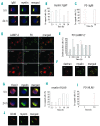
Design and methods: We used a well-characterized monoclonal gammopathy of undetermined significance-derived B-cell line, TJ2, expressing anti-protein zero immunoglobulin M. The ability of TJ2 cells to bind, endocytose, process, and present protein zero was investigated by receptor-clustering and immunofluorescence. The activation of protein zero-specific autologous T cells was studied by measuring interleukin-2 and interferon-gamma with flow cytometry, immunobeads, and enzyme-linked immunospot assays.
Results: Surface-receptor clustering and endocytosis of receptor-ligand (immunoglobulin M/protein zero) complexes were pronounced after exposure to protein zero. Naturally processed or synthetic protein zero peptide (194-208)-pulsed TJ2 cells significantly induced interleukin-2 secretion from autologous T cells compared to control antigen-pulsed cells (P<0.001). The numbers of interferon-gamma-producing T helper cells, including CD4(+)/CD8(+) cells, were also significantly increased (P=0.0152). Affinity-isolated naturally processed myelin peptides were potent interferon-gamma stimulators for autologous peripheral blood mononuclear cells, but not for control peripheral blood mononuclear cells.
Conclusions: We show for the first time that myelin protein zero is naturally processed in B cells from monoclonal gammopathy of undetermined significance of immunoglobulin M isotype, acting as aberrant antigen-presenting cells in activation of a patient's T helper cells. Our findings cast new light on the important role of autoreactive protein zero-specific B cells in the induction of the pathogenic T-cell responses found in nerve lesions of patients with monoclonal gammopathy of undetermined significance with peripheral neuropathy.
Figures


Similar articles
-
Myelin protein P0-specific IgM producing monoclonal B cell lines were established from polyneuropathy patients with monoclonal gammopathy of undetermined significance (MGUS).Clin Exp Immunol. 2002 Feb;127(2):255-62. doi: 10.1046/j.1365-2249.2002.01739.x. Clin Exp Immunol. 2002. PMID: 11876747 Free PMC article.
-
Secretions of anti-myelin-associated glycoprotein antibodies by B cells from patients with neuropathy and nonmalignant monoclonal gammopathy.Cell Immunol. 1986 Dec;103(2):434-40. doi: 10.1016/0008-8749(86)90103-6. Cell Immunol. 1986. PMID: 2433053
-
Autoreactive T and B cell responses to myelin antigens after diagnostic sural nerve biopsy.J Neurol Sci. 1993 Jul;117(1-2):130-9. doi: 10.1016/0022-510x(93)90165-u. J Neurol Sci. 1993. PMID: 7691993
-
Monoclonal gammopathy and neuropathy.Curr Opin Neurol. 1996 Oct;9(5):334-7. doi: 10.1097/00019052-199610000-00003. Curr Opin Neurol. 1996. PMID: 8894407 Review.
-
[Polyneuropathies and monoclonal gammopathies, so-called benign].Rev Prat. 1992 Jan 1;42(1):43-5. Rev Prat. 1992. PMID: 1314413 Review. French.
Cited by
-
Antibodies to myelin P0 and ceramide perpetuate neuropathy in long standing treated leprosy patients.Neurochem Res. 2011 May;36(5):766-73. doi: 10.1007/s11064-010-0397-7. Epub 2011 Jan 14. Neurochem Res. 2011. PMID: 21234675
-
Serological responses to prednisolone treatment in leprosy reactions: study of TNF-α, antibodies to phenolic glycolipid-1, lipoarabinomanan, ceramide and S100-B.Lipids Health Dis. 2014 Jul 28;13:119. doi: 10.1186/1476-511X-13-119. Lipids Health Dis. 2014. PMID: 25070345 Free PMC article.
References
-
- Kyle RA, Therneau TM, Rajkumar SV, Larson DR, Plevak MF, Offord JR, et al. Prevalence of monoclonal gammopathy of undetermined significance. N Engl J Med. 2006;354(13):1362–9. - PubMed
-
- Martín-Jiménez P, García-Sanz R, Balanzategui A, Alcoceba M, Ocio E, Sanchez ML, et al. Molecular characterization of heavy chain immunoglobulin gene rearrangements in Waldenström’s macroglobulinemia and IgM monoclonal gammopathy of undetermined significance. Haematologica. 2007;92(5):635–42. - PubMed
-
- Sahota SS, Forconi F, Ottensmeier CH, Provan D, Oscier DG, Hamblin TJ, Stevenson FK. Typical Waldenström macroglobulinemia is derived from a B-cell arrested after cessation of somatic mutation but prior to isotype switch events. Blood. 2002;100(4):1505–7. - PubMed
-
- Kyle RA, Therneau TM, Rajkumar SV, Offord JR, Larson DR, Plevak MF, Melton LJ., 3rd A long-term study of prognosis in monoclonal gammopathy of undetermined significance. N Engl J Med. 2002;346(8):564–9. - PubMed
-
- Baldini L, Goldaniga M, Guffanti A, Broglia C, Cortelazzo S, Rossi A, et al. Immunoglobulin M monoclonal gammopathies of undetermined significance and indolent Waldenström’s macroglobulinemia recognize the same determinants of evolution into symptomatic lymphoid disorders: proposal for a common prognostic scoring system. J Clin Oncol. 2005;23(21):4662–8. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

