Specific saposin C deficiency: CNS impairment and acid beta-glucosidase effects in the mouse
- PMID: 20015957
- PMCID: PMC2807372
- DOI: 10.1093/hmg/ddp531
Specific saposin C deficiency: CNS impairment and acid beta-glucosidase effects in the mouse
Abstract
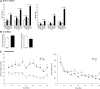
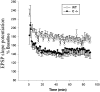
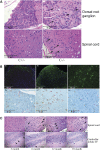
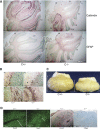
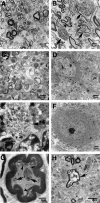
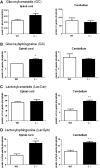
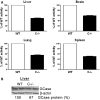
Saposins A, B, C and D are derived from a common precursor, prosaposin (psap). The few patients with saposin C deficiency develop a Gaucher disease-like central nervous system (CNS) phenotype attributed to diminished glucosylceramide (GC) cleavage activity by acid beta-glucosidase (GCase). The in vivo effects of saposin C were examined by creating mice with selective absence of saposin C (C-/-) using a knock-in point mutation (cysteine-to-proline) in exon 11 of the psap gene. In C-/- mice, prosaposin and saposins A, B and D proteins were present at near wild-type levels, but the saposin C protein was absent. By 1 year, the C-/- mice exhibited weakness of the hind limbs and progressive ataxia. Decreased neuromotor activity and impaired hippocampal long-term potentiation were evident. Foamy storage cells were observed in dorsal root ganglion and there was progressive loss of cerebellar Purkinje cells and atrophy of cerebellar granule cells. Ultrastructural analyses revealed inclusions in axonal processes in the spinal cord, sciatic nerve and brain, but no excess of multivesicular bodies. Activated microglial cells and astrocytes were present in thalamus, brain stem, cerebellum and spinal cord, indicating regional pro-inflammatory responses. No storage cells were found in visceral organs of these mice. The absence of saposin C led to moderate increases in GC and lactosylceramide (LacCer) and their deacylated analogues. These results support the view that saposin C has multiple roles in glycosphingolipid (GSL) catabolism as well as a prominent function in CNS and axonal integrity independent of its role as an optimizer/stabilizer of GCase.
Figures

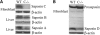

References
-
- Leonova T., Qi X., Bencosme A., Ponce E., Sun Y., Grabowski G.A. Proteolytic processing patterns of prosaposin in insect and mammalian cells. J. Biol. Chem. 1996;271:17312–17320. - PubMed
-
- Sandhoff K., Kolter T., Van Echten-Deckert G. Sphingolipid metabolism. Sphingoid analogs, sphingolipid activator proteins, and the pathology of the cell. Ann. NY Acad. Sci. 1998;845:139–151. - PubMed
-
- Li S.C., Sonnino S., Tettamanti G., Li Y.T. Characterization of a nonspecific activator protein for the enzymatic hydrolysis of glycolipids. J. Biol. Chem. 1988;263:6588–6591. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

