Transplanted neural precursors enhance host brain-derived myelin regeneration
- PMID: 20016084
- PMCID: PMC6666185
- DOI: 10.1523/JNEUROSCI.3364-09.2009
Transplanted neural precursors enhance host brain-derived myelin regeneration
Abstract
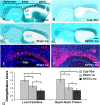
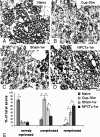
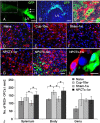
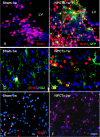
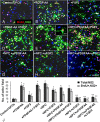
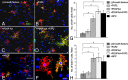
In multiple sclerosis lesions resident oligodendrocyte progenitor cells (OPCs) are present, but fail to remyelinate. In the current study we examined whether neural precursor cell (NPC) transplantation can facilitate host brain-derived remyelination. We used the chronic cuprizone-induced demyelination model in aged mice, in which slow remyelination follows cuprizone removal. NPCs were transplanted to the lateral ventricles (intracerebroventricular) of cuprizone-induced demyelinated brains. In this experimental setup, transplanted cells remained mostly in the periventricular area in an undifferentiated state. The extent of demyelination, remyelination, and proliferation of host brain regenerative cell population were examined at 1 week posttransplantation in the splenium of the corpus callosum, which was devoid of any transplanted cells. Transplantation of NPCs, but not of control, human embryonic kidney cells, significantly enhanced remyelination compared with sham-operated mice. Remyelination was performed exclusively by host brain OPCs. The proregenerative effect of transplanted NPCs was related to an increase in the proliferation of host brain OPCs. To examine the mechanism that underlies the proregenerative effect of NPCs in vitro, we used an NPC-OPC coculture system. These experiments indicated that NPCs induced the proliferation of OPCs and facilitated their differentiation into mature oligodendrocytes. The mitogenic effect of NPCs was mediated by platelet-derived growth factor-AA and fibroblast growth factor-2. In conclusion, NPC transplantation enhances host-derived myelin regeneration following chronic demyelination. This trophic effect may stimulate resident OPCs to overcome the remyelination failure in multiple sclerosis.
Figures
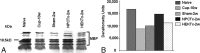
Similar articles
-
Adult neural precursor cells from the subventricular zone contribute significantly to oligodendrocyte regeneration and remyelination.J Neurosci. 2014 Oct 15;34(42):14128-46. doi: 10.1523/JNEUROSCI.3491-13.2014. J Neurosci. 2014. PMID: 25319708 Free PMC article.
-
Neural Stem Cells of the Subventricular Zone Contribute to Neuroprotection of the Corpus Callosum after Cuprizone-Induced Demyelination.J Neurosci. 2019 Jul 10;39(28):5481-5492. doi: 10.1523/JNEUROSCI.0227-18.2019. Epub 2019 May 28. J Neurosci. 2019. PMID: 31138656 Free PMC article.
-
Transplanted miR-219-overexpressing oligodendrocyte precursor cells promoted remyelination and improved functional recovery in a chronic demyelinated model.Sci Rep. 2017 Feb 1;7:41407. doi: 10.1038/srep41407. Sci Rep. 2017. PMID: 28145507 Free PMC article.
-
Matrix metalloproteinases shape the oligodendrocyte (niche) during development and upon demyelination.Neurosci Lett. 2020 Jun 11;729:134980. doi: 10.1016/j.neulet.2020.134980. Epub 2020 Apr 19. Neurosci Lett. 2020. PMID: 32315713 Review.
-
The role of oligodendrocytes and oligodendrocyte progenitors in CNS remyelination.Adv Exp Med Biol. 1999;468:183-97. doi: 10.1007/978-1-4615-4685-6_15. Adv Exp Med Biol. 1999. PMID: 10635029 Review.
Cited by
-
Forced Remyelination Promotes Axon Regeneration in a Rat Model of Spinal Cord Injury.Int J Mol Sci. 2022 Dec 28;24(1):495. doi: 10.3390/ijms24010495. Int J Mol Sci. 2022. PMID: 36613945 Free PMC article.
-
CXCL12-mediated guidance of migrating embryonic stem cell-derived neural progenitors transplanted into the hippocampus.PLoS One. 2010 Dec 31;5(12):e15856. doi: 10.1371/journal.pone.0015856. PLoS One. 2010. PMID: 21209827 Free PMC article.
-
PET molecular imaging in stem cell therapy for neurological diseases.Eur J Nucl Med Mol Imaging. 2011 Oct;38(10):1926-38. doi: 10.1007/s00259-011-1860-7. Epub 2011 Jun 23. Eur J Nucl Med Mol Imaging. 2011. PMID: 21698414 Review.
-
Aggregation of MBP in chronic demyelination.Ann Clin Transl Neurol. 2015 Jul;2(7):711-21. doi: 10.1002/acn3.207. Epub 2015 Jun 6. Ann Clin Transl Neurol. 2015. PMID: 26273684 Free PMC article.
-
The Molecular Basis for Remyelination Failure in Multiple Sclerosis.Cells. 2019 Aug 3;8(8):825. doi: 10.3390/cells8080825. Cells. 2019. PMID: 31382620 Free PMC article. Review.
References
-
- Ben-Hur T, Einstein O, Mizrachi-Kol R, Ben-Menachem O, Reinhartz E, Karussis D, Abramsky O. Transplanted multipotential neural precursor cells migrate into the inflamed white matter in response to experimental autoimmune encephalomyelitis. Glia. 2003;41:73–80. - PubMed
-
- Calver AR, Hall AC, Yu WP, Walsh FS, Heath JK, Betsholtz C, Richardson WD. Oligodendrocyte population dynamics and the role of PDGF in vivo. Neuron. 1998;20:869–882. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
