Direct interaction of GABAB receptors with M2 muscarinic receptors enhances muscarinic signaling
- PMID: 20016095
- PMCID: PMC2818375
- DOI: 10.1523/JNEUROSCI.4103-09.2009
Direct interaction of GABAB receptors with M2 muscarinic receptors enhances muscarinic signaling
Abstract
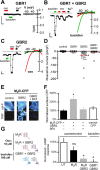
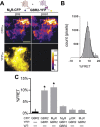
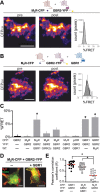
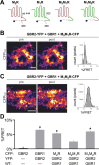
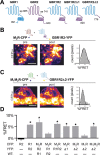
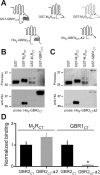
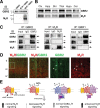
Downregulation of G-protein-coupled receptors (GPCRs) provides an important mechanism for reducing neurotransmitter signaling during sustained stimulation. Chronic stimulation of M(2) muscarinic receptors (M(2)Rs) causes internalization of M(2)R and G-protein-activated inwardly rectifying potassium (GIRK) channels in neuronal PC12 cells, resulting in loss of function. Here, we show that coexpression of GABA(B) R2 receptors (GBR2s) rescues both surface expression and function of M(2)R, including M(2)R-induced activation of GIRKs and inhibition of cAMP production. GBR2 showed significant association with M(2)R at the plasma membrane but not other GPCRs (M(1)R, mu-opioid receptor), as detected by fluorescence resonance energy transfer measured with total internal reflection fluorescence microscopy. Unique regions of the proximal C-terminal domains of GBR2 and M(2)R mediate specific binding between M(2)R and GBR2. In the brain, GBR2, but not GBR1, biochemically coprecipitates with M(2)R and overlaps with M(2)R expression in cortical neurons. This novel heteromeric association between M(2)R and GBR2 provides a possible mechanism for altering muscarinic signaling in the brain and represents a previously unrecognized role for GBR2.
Figures

References
-
- Blein S, Ginham R, Uhrin D, Smith BO, Soares DC, Veltel S, McIlhinney RA, White JH, Barlow PN. Structural analysis of the complement control protein (CCP) modules of GABA(B) receptor 1a: only one of the two CCP modules is compactly folded. J Biol Chem. 2004;279:48292–48306. - PubMed
-
- Calver AR, Michalovich D, Testa TT, Robbins MJ, Jaillard C, Hill J, Szekeres PG, Charles KJ, Jourdain S, Holbrook JD, Boyfield I, Patel N, Medhurst AD, Pangalos MN. Molecular cloning and characterisation of a novel GABAB-related G-protein coupled receptor. Brain Res Mol Brain Res. 2003;110:305–317. - PubMed
-
- Chang W, Tu C, Cheng Z, Rodriguez L, Chen TH, Gassmann M, Bettler B, Margeta M, Jan LY, Shoback D. Complex formation with the type B gamma-aminobutyric acid receptor affects the expression and signal transduction of the extracellular calcium-sensing receptor. Studies with HEK-293 cells and neurons. J Biol Chem. 2007;282:25030–25040. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- NS056359/NS/NINDS NIH HHS/United States
- R01 NS048045/NS/NINDS NIH HHS/United States
- NS048045/NS/NINDS NIH HHS/United States
- NS051195/NS/NINDS NIH HHS/United States
- DA019022/DA/NIDA NIH HHS/United States
- R21 NS080064/NS/NINDS NIH HHS/United States
- R01 NS051195/NS/NINDS NIH HHS/United States
- NS054900/NS/NINDS NIH HHS/United States
- P01 NS054900/NS/NINDS NIH HHS/United States
- R01 DA019022/DA/NIDA NIH HHS/United States
- R01 NS037682/NS/NINDS NIH HHS/United States
- R01 NS056359/NS/NINDS NIH HHS/United States
- R01 NS081986/NS/NINDS NIH HHS/United States
- NS047478/NS/NINDS NIH HHS/United States
- R01 NS047478/NS/NINDS NIH HHS/United States
- NS37682/NS/NINDS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
