Apyrase (nucleoside triphosphate-diphosphohydrolase) and extracellular nucleotides regulate cotton fiber elongation in cultured ovules
- PMID: 20018604
- PMCID: PMC2815863
- DOI: 10.1104/pp.109.147637
Apyrase (nucleoside triphosphate-diphosphohydrolase) and extracellular nucleotides regulate cotton fiber elongation in cultured ovules
Abstract
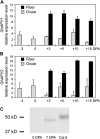
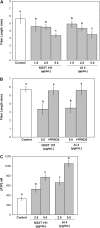
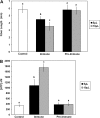
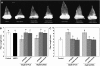
Ectoapyrase enzymes remove the terminal phosphate from extracellular nucleoside tri- and diphosphates. In Arabidopsis (Arabidopsis thaliana), two ectoapyrases, AtAPY1 and AtAPY2, have been implicated as key modulators of growth. In fibers of cotton (Gossypium hirsutum), transcript levels for GhAPY1 and GhAPY2, two closely related ectoapyrases that have high sequence similarity to AtAPY1 and AtAPY2, are up-regulated when fibers enter their rapid growth phase. In an ovule culture system, fibers release ATP as they grow, and when their ectoapyrase activity is blocked by the addition of polyclonal anti-apyrase antibodies or by two different small molecule inhibitors, the medium ATP level rises and fiber growth is suppressed. High concentrations of the poorly hydrolyzable nucleotides ATPgammaS and ADPbetaS applied to the medium inhibit fiber growth, and low concentrations of them stimulate growth, but treatment with adenosine 5'-O-thiomonophosphate causes no change in the growth rate. Both the inhibition and stimulation of growth by applied nucleotides can be blocked by an antagonist that blocks purinoceptors in animal cells, and by adenosine. Treatment of cotton ovule cultures with ATPgammaS induces increased levels of ethylene, and two ethylene antagonists, aminovinylglycine and silver nitrate, block both the growth stimulatory and growth inhibitory effects of applied nucleotides. In addition, the ethylene precursor, 1-aminocyclopropane-1-carboxylic acid, lowers the concentration of nucleotide needed to promote fiber growth. These data indicate that ectoapyrases and extracellular nucleotides play a significant role in regulating cotton fiber growth and that ethylene is a likely downstream component of the signaling pathway.
Figures
References
-
- Bendtsen JD, Nielsen H, von Heijne G, Brunak S (2004) Improved prediction of signal peptides: SignalP 3.0. J Mol Biol 340 783–795 - PubMed
-
- Bengtsson T, Zalavary S, Stendahl O, Grenegard M (1996) Release of oxygen metabolites from chemoattractant-stimulated neutrophils is inhibited by resting platelets: role of extracellular adenosine and actin polymerization. Blood 87 4411–4423 - PubMed
-
- Basra AS, Malik CP (1984) Development of the cotton fiber. Int Rev Cytol 89 65–113
-
- Burnstock G (1996) Purinoceptors: ontogeny and phylogeny. Drug Dev Res 39 204–242
-
- Burnstock G (2008) Purinergic signalling and disorders of the central nervous system. Nat Rev Drug Discov 7 575–590 - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

