Mice with a selective impairment of IFN-gamma signaling in macrophage lineage cells demonstrate the critical role of IFN-gamma-activated macrophages for the control of protozoan parasitic infections in vivo
- PMID: 20018611
- PMCID: PMC2886308
- DOI: 10.4049/jimmunol.0902346
Mice with a selective impairment of IFN-gamma signaling in macrophage lineage cells demonstrate the critical role of IFN-gamma-activated macrophages for the control of protozoan parasitic infections in vivo
Abstract
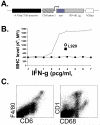
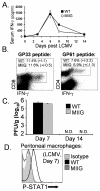
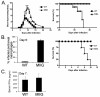
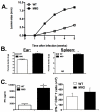
IFN-gamma has long been recognized as a cytokine with potent and varied effects in the immune response. Although its effects on specific cell types have been well studied in vitro, its in vivo effects are less clearly understood because of its diverse actions on many different cell types. Although control of multiple protozoan parasites is thought to depend critically on the direct action of IFN-gamma on macrophages, this premise has never been directly proven in vivo. To more directly examine the effects of IFN-gamma on cells of the macrophage lineage in vivo, we generated mice called the "macrophages insensitive to IFN-gamma" (MIIG) mice, which express a dominant negative mutant IFN-gamma receptor in CD68+ cells: monocytes, macrophages, dendritic cells, and mast cells. Macrophage lineage cells and mast cells from these mice are unable to respond to IFN-gamma, whereas other cells are able to produce and respond to this cytokine normally. When challenged in vitro, macrophages from MIIG mice were unable produce NO or kill Trypanosoma cruzi or Leishmania major after priming with IFN-gamma. Furthermore, MIIG mice demonstrated impaired parasite control and heightened mortality after T. cruzi, L. major, and Toxoplasma gondii infection, despite an appropriate IFN-gamma response. In contrast, MIIG mice displayed normal control of lymphocytic choriomeningitis virus, despite persistent insensitivity of macrophages to IFN-gamma. Thus, the MIIG mouse formally demonstrates for the first time in vivo, the specific importance of direct, IFN-gamma mediated activation of macrophages for controlling infection with multiple protozoan parasites.
Figures


References
-
- Bach EA, Aguet M, Schreiber RD. The IFN gamma receptor: a paradigm for cytokine receptor signaling. Annu Rev Immunol. 1997;15:563–591. - PubMed
-
- Pestka S, Krause CD, Walter MR. Interferons, interferon-like cytokines, and their receptors. Immunol Rev. 2004;202:8–32. - PubMed
-
- Strehl B, Seifert U, Kruger E, Heink S, Kuckelkorn U, Kloetzel PM. Interferon-gamma, the functional plasticity of the ubiquitin-proteasome system, and MHC class I antigen processing. Immunol Rev. 2005;207:19–30. - PubMed
-
- Muhl H, Pfeilschifter J. Anti-inflammatory properties of pro-inflammatory interferon-gamma. Int Immunopharmacol. 2003;3:1247–1255. - PubMed
-
- Badovinac VP, Tvinnereim AR, Harty JT. Regulation of antigen-specific CD8+ T cell homeostasis by perforin and interferon-gamma. Science. 2000;290:1354–1358. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

