Embryonic stem cell tumor model reveals role of vascular endothelial receptor tyrosine phosphatase in regulating Tie2 pathway in tumor angiogenesis
- PMID: 20018779
- PMCID: PMC2794035
- DOI: 10.1073/pnas.0911189106
Embryonic stem cell tumor model reveals role of vascular endothelial receptor tyrosine phosphatase in regulating Tie2 pathway in tumor angiogenesis
Abstract
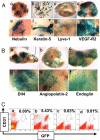
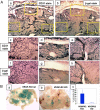
Inhibiting angiogenesis has become an effective approach for treating cancer and other diseases. However, our understanding of signaling pathways in tumor angiogenesis has been limited by the embryonic lethality of many gene knockouts. To overcome this limitation, we used the plasticity of embryonic stem (ES) cells to develop a unique approach to study tumor angiogenesis. Murine ES cells can be readily manipulated genetically; in addition, ES cells implanted subcutaneously in mice develop into tumors that contain a variety of cell types (teratomas). We show that ES cells differentiate into bona fide endothelial cells within the teratoma, and that these ES-derived endothelial cells form part of the functional tumor vasculature. Using this powerful and flexible system, the Angiopoietin/Tie2 system is shown to have a key role in the regulation of tumor vessel size. Endothelial differentiation in the ES teratoma model allows gene-targeting methods to be used in the study of tumor angiogenesis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Differential inhibition of tumor angiogenesis by tie2 and vascular endothelial growth factor receptor-2 dominant-negative receptor mutants.Int J Cancer. 2001 Feb 1;91(3):273-82. doi: 10.1002/1097-0215(200002)9999:9999<::aid-ijc1054>3.0.co;2-q. Int J Cancer. 2001. PMID: 11169947
-
Platelet-derived growth factor receptor-beta constitutive activity promotes angiogenesis in vivo and in vitro.Arterioscler Thromb Vasc Biol. 2007 Oct;27(10):2142-9. doi: 10.1161/01.ATV.0000282198.60701.94. Epub 2007 Jul 26. Arterioscler Thromb Vasc Biol. 2007. PMID: 17656670
-
Beta 1 integrin is essential for teratoma growth and angiogenesis.J Cell Biol. 1997 Oct 6;139(1):265-78. doi: 10.1083/jcb.139.1.265. J Cell Biol. 1997. PMID: 9314545 Free PMC article.
-
Tie-1: A potential target for anti-angiogenesis therapy.J Huazhong Univ Sci Technolog Med Sci. 2015 Oct;35(5):615-622. doi: 10.1007/s11596-015-1479-1. Epub 2015 Oct 22. J Huazhong Univ Sci Technolog Med Sci. 2015. PMID: 26489611 Review.
-
Functional significance of Tie2 signaling in the adult vasculature.Recent Prog Horm Res. 2004;59:51-71. doi: 10.1210/rp.59.1.51. Recent Prog Horm Res. 2004. PMID: 14749497 Review.
Cited by
-
Similarities and differences in the regulation of leukocyte extravasation and vascular permeability.Semin Immunopathol. 2014 Mar;36(2):177-92. doi: 10.1007/s00281-014-0419-7. Epub 2014 Mar 18. Semin Immunopathol. 2014. PMID: 24638889 Review.
-
Angiopoietin-1 deficiency increases tumor metastasis in mice.BMC Cancer. 2017 Aug 11;17(1):539. doi: 10.1186/s12885-017-3531-y. BMC Cancer. 2017. PMID: 28800750 Free PMC article.
-
Effects of vascular-endothelial protein tyrosine phosphatase inhibition on breast cancer vasculature and metastatic progression.J Natl Cancer Inst. 2013 Aug 21;105(16):1188-201. doi: 10.1093/jnci/djt164. Epub 2013 Jul 30. J Natl Cancer Inst. 2013. PMID: 23899555 Free PMC article.
-
Embryonic Stem Cell-Like Subpopulations in Venous Malformation.Front Med (Lausanne). 2017 Oct 4;4:162. doi: 10.3389/fmed.2017.00162. eCollection 2017. Front Med (Lausanne). 2017. PMID: 29046873 Free PMC article.
-
The Tie2 signaling pathway in retinal vascular diseases: a novel therapeutic target in the eye.Int J Retina Vitreous. 2020 Oct 13;6:48. doi: 10.1186/s40942-020-00250-z. eCollection 2020. Int J Retina Vitreous. 2020. PMID: 33072401 Free PMC article. Review.
References
-
- Ferrara N, Kerbel RS. Angiogenesis as a therapeutic target. Nature. 2005;438:967–974. - PubMed
-
- Folkman J. The role of angiogenesis in tumor growth. Semin Cancer Biol. 1992;3:65–71. - PubMed
-
- Robertson EJ. Using embryonic stem cells to introduce mutations into the mouse germ line. Biol Reprod. 1991;44:238–245. - PubMed
-
- Shalaby F, et al. Failure of blood-island formation and vasculogenesis in Flk-1-deficient mice. Nature. 1995;376:62–66. - PubMed
-
- Ferrara N, et al. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature. 1996;380:439–442. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

