Vestibuloocular reflex adaptation investigated with chronic motion-modulated electrical stimulation of semicircular canal afferents
- PMID: 20018838
- PMCID: PMC2822686
- DOI: 10.1152/jn.00241.2009
Vestibuloocular reflex adaptation investigated with chronic motion-modulated electrical stimulation of semicircular canal afferents
Abstract
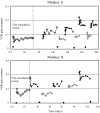
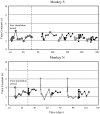
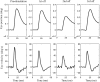
To investigate vestibuloocular reflex (VOR) adaptation produced by changes in peripheral vestibular afference, we developed and tested a vestibular "prosthesis" that senses yaw-axis angular head velocity and uses this information to modulate the rate of electrical pulses applied to the lateral canal ampullary nerve. The ability of the brain to adapt the different components of the VOR (gain, phase, axis, and symmetry) during chronic prosthetic electrical stimulation was studied in two squirrel monkeys. After characterizing the normal yaw-axis VOR, electrodes were implanted in both lateral canals and the canals were plugged. The VOR in the canal-plugged/instrumented state was measured and then unilateral stimulation was applied by the prosthesis. The VOR was repeatedly measured over several months while the prosthetic stimulation was cycled between off, low-sensitivity, and high-sensitivity stimulation states. The VOR response initially demonstrated a low gain, abnormal rotational axis, and substantial asymmetry. During chronic stimulation the gain increased, the rotational axis improved, and the VOR became more symmetric. Gain changes were augmented by cycling the stimulation between the off and both low- and high-sensitivity states every few weeks. The VOR time constant remained low throughout the period of chronic stimulation. These results demonstrate that the brain can adaptively modify the gain, axis, and symmetry of the VOR when provided with chronic motion-modulated electrical stimulation by a canal prosthesis.
Figures





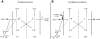
 ” through the canal ampullary nerve. I, type I vestibular neurons; II, type II vestibular neurons; OMN, ocular motor neurons; PC, Purkinje cells. Black arrows represent excitatory connections and gray arrows inhibitory connections.
” through the canal ampullary nerve. I, type I vestibular neurons; II, type II vestibular neurons; OMN, ocular motor neurons; PC, Purkinje cells. Black arrows represent excitatory connections and gray arrows inhibitory connections.Similar articles
-
Vestibular adaptation studied with a prosthetic semicircular canal.J Vestib Res. 2002-2003;12(2-3):87-94. J Vestib Res. 2002. PMID: 12867667
-
Effects of canal plugging on the vestibuloocular reflex and vestibular nerve discharge during passive and active head rotations.J Neurophysiol. 2009 Nov;102(5):2693-703. doi: 10.1152/jn.00710.2009. Epub 2009 Sep 2. J Neurophysiol. 2009. PMID: 19726724 Free PMC article.
-
Spatial and temporal properties of eye movements produced by electrical stimulation of semicircular canal afferents.J Neurophysiol. 2012 Sep;108(5):1511-20. doi: 10.1152/jn.01029.2011. Epub 2012 Jun 6. J Neurophysiol. 2012. PMID: 22673321 Free PMC article.
-
Adaptive plasticity in the otolith-ocular reflex.Auris Nasus Larynx. 2003 Feb;30 Suppl:S3-6. doi: 10.1016/s0385-8146(02)00117-7. Auris Nasus Larynx. 2003. PMID: 12543152 Review.
-
Ontogenetic Development of Vestibulo-Ocular Reflexes in Amphibians.Front Neural Circuits. 2016 Nov 8;10:91. doi: 10.3389/fncir.2016.00091. eCollection 2016. Front Neural Circuits. 2016. PMID: 27877114 Free PMC article. Review.
Cited by
-
The Vestibular Implant Input Interacts with Residual Natural Function.Front Neurol. 2017 Dec 14;8:644. doi: 10.3389/fneur.2017.00644. eCollection 2017. Front Neurol. 2017. PMID: 29312107 Free PMC article.
-
Advances in the diagnosis and treatment of vestibular disorders: psychophysics and prosthetics.J Neurosci. 2015 Apr 1;35(13):5089-96. doi: 10.1523/JNEUROSCI.3922-14.2015. J Neurosci. 2015. PMID: 25834036 Free PMC article. Review.
-
Simultaneous activation of multiple vestibular pathways upon electrical stimulation of semicircular canal afferents.J Neurol. 2020 Dec;267(Suppl 1):273-284. doi: 10.1007/s00415-020-10120-1. Epub 2020 Aug 10. J Neurol. 2020. PMID: 32778921 Free PMC article.
-
Vestibular assistance systems: promises and challenges.J Neurol. 2016 Apr;263 Suppl 1:S30-5. doi: 10.1007/s00415-015-7922-1. Epub 2016 Apr 15. J Neurol. 2016. PMID: 27083882 Free PMC article. Review.
-
Cross-axis adaptation improves 3D vestibulo-ocular reflex alignment during chronic stimulation via a head-mounted multichannel vestibular prosthesis.Exp Brain Res. 2011 May;210(3-4):595-606. doi: 10.1007/s00221-011-2591-5. Epub 2011 Mar 4. Exp Brain Res. 2011. PMID: 21374081 Free PMC article.
References
-
- Agrawal Y, Carey JP, Della Santina CC, Schubert MC, Minor LB. Disorders of balance and vestibular function in US adults: data from the National Health and Nutritional Examination Survey, 2001–2004. Arch Intern Med 169: 938–944, 2009 - PubMed
-
- Angelaki DE, Hess BJM, Arai Y, Suzuki J-I. Adaptation of primate vestibuloocular reflex to altered peripheral vestibular inputs. I. Frequency-specific recovery of horizontal VOR after inactivation of the lateral semicircular canals. J Neurophysiol 76: 2941–2953, 1996 - PubMed
-
- Blanks RH, Curthoys IS, Bennett ML, Markham CH. Planar relationship of the semicircular canals in rhesus and squirrel monkeys. Brain Res 340: 315–324, 1985 - PubMed
-
- Broussard DM, Bhatia JK, Jones GEG. The dynamics of the vestibulo-ocular reflex after peripheral vestibular damage. I. Frequency-dependent asymmetry. Exp Brain Res 125: 353–364, 1999 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

