Cyclic mechanical stretch decreases cell migration by inhibiting phosphatidylinositol 3-kinase- and focal adhesion kinase-mediated JNK1 activation
- PMID: 20018857
- PMCID: PMC2836056
- DOI: 10.1074/jbc.M109.084335
Cyclic mechanical stretch decreases cell migration by inhibiting phosphatidylinositol 3-kinase- and focal adhesion kinase-mediated JNK1 activation
Abstract
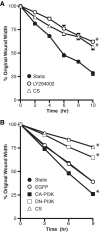
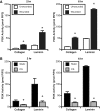
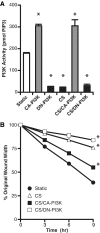
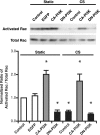
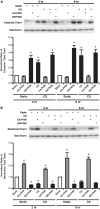
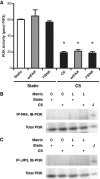
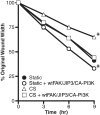
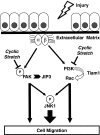
Epithelial cell migration during wound healing requires coordinated signaling pathways that direct polarization of the leading and trailing ends of the cells, cytoskeletal organization, and remodeling of focal adhesions. These inherently mechanical processes are disrupted by cyclic stretch (CS), but the specific signaling molecules involved in this disruption are not well understood. In this study, we demonstrate that inhibition of phosphatidylinositol 3-kinase (PI3K) or expression of a dominant-negative form of PI3K caused inhibition of airway epithelial cell wound closure. CS caused a sustained decrease in activation of PI3K and inhibited wound healing. Expression of constitutively active PI3K stimulated translocation of Tiam1 to the membrane, increased Rac1 activity, and increased wound healing of airway epithelial cells. Increased Rac1 activity resulted in increased phosphorylation of JNK1. PI3K activation was not regulated by association with focal adhesion kinase. Restoration of efficient cell migration during CS required coexpression of constitutively active PI3K, focal adhesion kinase, and JIP3.
Figures

Similar articles
-
Mechanical stretch decreases FAK phosphorylation and reduces cell migration through loss of JIP3-induced JNK phosphorylation in airway epithelial cells.Am J Physiol Lung Cell Mol Physiol. 2009 Sep;297(3):L520-9. doi: 10.1152/ajplung.00076.2009. Epub 2009 Jul 2. Am J Physiol Lung Cell Mol Physiol. 2009. PMID: 19574423 Free PMC article.
-
Mechanical stretch decreases migration of alveolar epithelial cells through mechanisms involving Rac1 and Tiam1.Am J Physiol Lung Cell Mol Physiol. 2008 Nov;295(5):L958-65. doi: 10.1152/ajplung.90218.2008. Epub 2008 Sep 19. Am J Physiol Lung Cell Mol Physiol. 2008. PMID: 18805958 Free PMC article.
-
The role of focal adhesion kinase-phosphatidylinositol 3-kinase-akt signaling in hepatic stellate cell proliferation and type I collagen expression.J Biol Chem. 2003 Mar 7;278(10):8083-90. doi: 10.1074/jbc.M212927200. Epub 2002 Dec 26. J Biol Chem. 2003. PMID: 12502711
-
Emodin inhibits tumor cell migration through suppression of the phosphatidylinositol 3-kinase-Cdc42/Rac1 pathway.Cell Mol Life Sci. 2005 May;62(10):1167-75. doi: 10.1007/s00018-005-5050-2. Cell Mol Life Sci. 2005. PMID: 15928809 Free PMC article.
-
The guanine nucleotide exchange factor Tiam1: a Janus-faced molecule in cellular signaling.Cell Signal. 2014 Mar;26(3):483-91. doi: 10.1016/j.cellsig.2013.11.034. Epub 2013 Dec 2. Cell Signal. 2014. PMID: 24308970 Review.
Cited by
-
The Role of PI3K in Met Driven Cancer: A Recap.Front Mol Biosci. 2018 Oct 24;5:86. doi: 10.3389/fmolb.2018.00086. eCollection 2018. Front Mol Biosci. 2018. PMID: 30406111 Free PMC article. Review.
-
Kymographic Imaging of the Elastic Modulus of Epithelial Cells during the Onset of Migration.Biophys J. 2015 Nov 17;109(10):2051-7. doi: 10.1016/j.bpj.2015.10.005. Biophys J. 2015. PMID: 26588564 Free PMC article.
-
Myosin-II-mediated directional migration of Dictyostelium cells in response to cyclic stretching of substratum.Biophys J. 2013 Feb 19;104(4):748-58. doi: 10.1016/j.bpj.2013.01.005. Biophys J. 2013. PMID: 23442953 Free PMC article.
-
Akt directly regulates focal adhesion kinase through association and serine phosphorylation: implication for pressure-induced colon cancer metastasis.Am J Physiol Cell Physiol. 2011 Mar;300(3):C657-70. doi: 10.1152/ajpcell.00377.2010. Epub 2011 Jan 5. Am J Physiol Cell Physiol. 2011. PMID: 21209368 Free PMC article.
-
A Fully Integrated Arduino-Based System for the Application of Stretching Stimuli to Living Cells and Their Time-Lapse Observation: A Do-It-Yourself Biology Approach.Ann Biomed Eng. 2021 Sep;49(9):2243-2259. doi: 10.1007/s10439-021-02758-3. Epub 2021 Mar 16. Ann Biomed Eng. 2021. PMID: 33728867
References
-
- Erjefält J. S., Erjefält I., Sundler F., Persson C. G. (1995) Cell Tissue Res. 281, 305–316 - PubMed
-
- Rickard K. A., Taylor J., Rennard S. I., Spurzem J. R. (1993) Am. J. Respir. Cell Mol. Biol. 8, 63–68 - PubMed
-
- White S. R., Dorscheid D. R., Rabe K. F., Wojcik K. R., Hamann K. J. (1999) Am. J. Respir. Cell Mol. Biol. 20, 787–796 - PubMed
-
- Cox B. D., Natarajan M., Stettner M. R., Gladson C. L. (2006) J. Cell. Biochem. 99, 35–52 - PubMed
-
- Schlaepfer D. D., Mitra S. K. (2004) Curr. Opin. Genet. Dev. 14, 92–101 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

