Mammalian target of rapamycin (mTOR) induces proliferation and de-differentiation responses to three coordinate pathophysiologic stimuli (mechanical strain, hypoxia, and extracellular matrix remodeling) in rat bladder smooth muscle
- PMID: 20019183
- PMCID: PMC2797892
- DOI: 10.2353/ajpath.2010.080834
Mammalian target of rapamycin (mTOR) induces proliferation and de-differentiation responses to three coordinate pathophysiologic stimuli (mechanical strain, hypoxia, and extracellular matrix remodeling) in rat bladder smooth muscle
Abstract
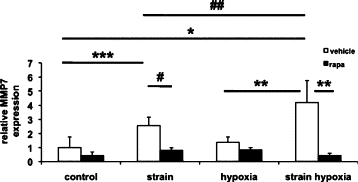
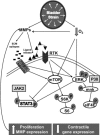
Maladaptive bladder muscle overgrowth and de-differentiation in human bladder obstructive conditions is instigated by coordinate responses to three stimuli: mechanical strain, tissue hypoxia, and extracellular matrix remodeling.( 1,2) Pathway analysis of genes induced by obstructive models of injury in bladder smooth muscle cells (BSMCs) identified a mammalian target of rapamycin (mTOR)-specific inhibitor as a potential pharmacological inhibitor. Strain-induced mTOR-specific S6K activation segregated differently from ERK1/2 activation in intact bladder ex vivo. Though rapamycin's antiproliferative effects in vascular smooth muscle cells are well known, its effects on BSMCs were previously unknown. Rapamycin significantly inhibited proliferation of BSMCs in response to mechanical strain, hypoxia, and denatured collagen. Rapamycin inhibited S6K at mTOR-sensitive phosphorylation sites in response to strain and hypoxia. Rapamycin also supported smooth muscle actin expression in response to strain or hypoxia-induced de-differentiation. Importantly, strain plus hypoxia synergistically augmented mTOR-dependent S6K activation, Mmp7 expression and proliferation. Forced expression of wild-type and constitutively active S6K resulted in loss of smooth muscle actin expression. Decreased smooth muscle actin, increased Mmp7 levels and mTOR pathway activation during in vivo partial bladder obstruction paralleled our in vitro studies. These results point to a coordinate role for mTOR in BSMCs responses to the three stimuli and a potential new therapeutic target for myopathic bladder disease.
Figures



Similar articles
-
Enhanced expression of glucose transporter-1 in vascular smooth muscle cells via the Akt/tuberous sclerosis complex subunit 2 (TSC2)/mammalian target of rapamycin (mTOR)/ribosomal S6 protein kinase (S6K) pathway in experimental renal failure.J Vasc Surg. 2013 Feb;57(2):475-85. doi: 10.1016/j.jvs.2012.07.037. Epub 2012 Dec 21. J Vasc Surg. 2013. PMID: 23265586
-
FAK mediates the activation of cardiac fibroblasts induced by mechanical stress through regulation of the mTOR complex.Cardiovasc Res. 2010 Jun 1;86(3):421-31. doi: 10.1093/cvr/cvp416. Epub 2009 Dec 28. Cardiovasc Res. 2010. Retraction in: Cardiovasc Res. 2014 Dec 1;104(3):512. doi: 10.1093/cvr/cvu228. PMID: 20038548 Retracted.
-
Mechanotransduction of extracellular signal-regulated kinases 1 and 2 mitogen-activated protein kinase activity in smooth muscle is dependent on the extracellular matrix and regulated by matrix metalloproteinases.Am J Pathol. 2006 Aug;169(2):459-70. doi: 10.2353/ajpath.2006.050969. Am J Pathol. 2006. PMID: 16877348 Free PMC article.
-
mTOR and vascular remodeling in lung diseases: current challenges and therapeutic prospects.FASEB J. 2013 May;27(5):1796-807. doi: 10.1096/fj.12-222224. Epub 2013 Jan 25. FASEB J. 2013. PMID: 23355268 Free PMC article. Review.
-
Common corruption of the mTOR signaling network in human tumors.Oncogene. 2008 Dec;27 Suppl 2(0 2):S43-51. doi: 10.1038/onc.2009.352. Oncogene. 2008. PMID: 19956179 Free PMC article. Review.
Cited by
-
Pathologic bladder microenvironment attenuates smooth muscle differentiation of skin derived precursor cells: implications for tissue regeneration.PLoS One. 2013;8(4):e59413. doi: 10.1371/journal.pone.0059413. Epub 2013 Apr 1. PLoS One. 2013. PMID: 23560047 Free PMC article.
-
Uropathogenic E. coli (UPEC) Infection Induces Proliferation through Enhancer of Zeste Homologue 2 (EZH2).PLoS One. 2016 Mar 10;11(3):e0149118. doi: 10.1371/journal.pone.0149118. eCollection 2016. PLoS One. 2016. PMID: 26964089 Free PMC article.
-
Adaptive inflammatory microenvironment for cell-based regeneration in ischemic cardiovascular disease.Organogenesis. 2013 Jul-Sep;9(3):121-4. doi: 10.4161/org.25586. Epub 2013 Jul 3. Organogenesis. 2013. PMID: 23974172 Free PMC article.
-
hASC and DFAT, Multipotent Stem Cells for Regenerative Medicine: A Comparison of Their Potential Differentiation In Vitro.Int J Mol Sci. 2017 Dec 13;18(12):2699. doi: 10.3390/ijms18122699. Int J Mol Sci. 2017. PMID: 29236047 Free PMC article.
-
Non-invasive voiding assessment in conscious mice.Bladder (San Franc). 2018 Apr 2;5(2):e33. doi: 10.14440/bladder.2018.582. eCollection 2018. Bladder (San Franc). 2018. PMID: 32775475 Free PMC article.
References
-
- Mattiasson A, Uvelius B. Changes in contractile properties in hypertrophic rat urinary bladder. J Urol. 1982;128:1340–1342. - PubMed
-
- Becker A, Baum M. Obstructive uropathy. Early Hum Dev. 2006;82:15–22. - PubMed
-
- Austin JC, Chacko SK, DiSanto M, Canning DA, Zderic SA. A male murine model of partial bladder outlet obstruction reveals changes in detrusor morphology, contractility and Myosin isoform expression. J Urol. 2004;172:1524–1528. - PubMed
-
- Buttyan R, Chen MW, Levin RM. Animal models of bladder outlet obstruction and molecular insights into the basis for the development of bladder dysfunction. Eur Urol. 1997;32 Suppl 1:32–39. - PubMed
-
- Johansson R, Persson K. Phenotypic modulation of cultured bladder smooth muscle cells and the expression of inducible nitric oxide synthase. Am J Physiol Regul Integr Comp Physiol. 2004;286:R642–R648. - PubMed
Uncited reference
-
- Locatelli F, Roger S. Comparative testing and pharmacovigilance of biosimilars. Nephrol, Dial, Transplant. 2006;21 Suppl 5:v13–v16. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

