Mechanical breaking of microtubules in axons during dynamic stretch injury underlies delayed elasticity, microtubule disassembly, and axon degeneration
- PMID: 20019243
- PMCID: PMC2879950
- DOI: 10.1096/fj.09-142844
Mechanical breaking of microtubules in axons during dynamic stretch injury underlies delayed elasticity, microtubule disassembly, and axon degeneration
Abstract
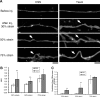
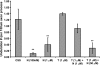
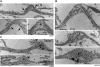
Little is known about which components of the axonal cytoskeleton might break during rapid mechanical deformation, such as occurs in traumatic brain injury. Here, we micropatterned neuronal cell cultures on silicone membranes to induce dynamic stretch exclusively of axon fascicles. After stretch, undulating distortions formed along the axons that gradually relaxed back to a straight orientation, demonstrating a delayed elastic response. Subsequently, swellings developed, leading to degeneration of almost all axons by 24 h. Stabilizing the microtubules with taxol maintained the undulating geometry after injury but greatly reduced axon degeneration. Conversely, destabilizing microtubules with nocodazole prevented undulations but greatly increased the rate of axon loss. Ultrastructural analyses of axons postinjury revealed immediate breakage and buckling of microtubules in axon undulations and progressive loss of microtubules. Collectively, these data suggest that dynamic stretch of axons induces direct mechanical failure at specific points along microtubules. This microtubule disorganization impedes normal relaxation of the axons, resulting in undulations. However, this physical damage also triggers progressive disassembly of the microtubules around the breakage points. While the disintegration of microtubules allows delayed recovery of the "normal" straight axon morphology, it comes at a great cost by interrupting axonal transport, leading to axonal swelling and degeneration.
Figures




References
-
- Gennarelli T A, Thibault L E, Adams J H, Graham D I, Thompson C J, Marcincin R P. Diffuse axonal injury and traumatic coma in the primate. Ann Neurol. 1982;12:564–574. - PubMed
-
- Smith D H, Meaney D F. Axonal damage in traumatic brain injury. Neuroscientist. 2000;6:483–495.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

