BCA2/Rabring7 promotes tetherin-dependent HIV-1 restriction
- PMID: 20019814
- PMCID: PMC2788703
- DOI: 10.1371/journal.ppat.1000700
BCA2/Rabring7 promotes tetherin-dependent HIV-1 restriction
Abstract
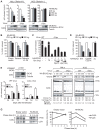
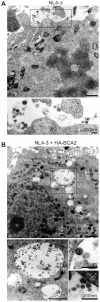
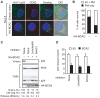
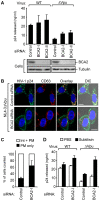
Host cell factors can either positively or negatively regulate the assembly and egress of HIV-1 particles from infected cells. Recent reports have identified a previously uncharacterized transmembrane protein, tetherin/CD317/BST-2, as a crucial host restriction factor that acts during a late budding step in HIV-1 replication by inhibiting viral particle release. Although tetherin has been shown to promote the retention of nascent viral particles on the host cell surface, the precise molecular mechanisms that occur during and after these tethering events remain largely unknown. We here report that a RING-type E3 ubiquitin ligase, BCA2 (Breast cancer-associated gene 2; also called Rabring7, ZNF364 or RNF115), is a novel tetherin-interacting host protein that facilitates the restriction of HIV-1 particle production in tetherin-positive cells. The expression of human BCA2 in "tetherin-positive" HeLa, but not in "tetherin-negative" HOS cells, resulted in a strong restriction of HIV-1 particle production. Upon the expression of tetherin in HOS cells, BCA2 was capable of inhibiting viral particle production as in HeLa cells. The targeted depletion of endogenous BCA2 by RNA interference (RNAi) in HeLa cells reduced the intracellular accumulation of viral particles, which were nevertheless retained on the plasma membrane. BCA2 was also found to facilitate the internalization of HIV-1 virions into CD63(+) intracellular vesicles leading to their lysosomal degradation. These results indicate that BCA2 accelerates the internalization and degradation of viral particles following their tethering to the cell surface and is a co-factor or enhancer for the tetherin-dependent restriction of HIV-1 release from infected cells.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
BCA2/Rabring7 targets HIV-1 Gag for lysosomal degradation in a tetherin-independent manner.PLoS Pathog. 2014 May 22;10(5):e1004151. doi: 10.1371/journal.ppat.1004151. eCollection 2014 May. PLoS Pathog. 2014. PMID: 24852021 Free PMC article.
-
The RING-CH ligase K5 antagonizes restriction of KSHV and HIV-1 particle release by mediating ubiquitin-dependent endosomal degradation of tetherin.PLoS Pathog. 2010 Apr 15;6(4):e1000843. doi: 10.1371/journal.ppat.1000843. PLoS Pathog. 2010. PMID: 20419159 Free PMC article.
-
Direct restriction of virus release and incorporation of the interferon-induced protein BST-2 into HIV-1 particles.PLoS Pathog. 2010 Mar 5;6(3):e1000701. doi: 10.1371/journal.ppat.1000701. PLoS Pathog. 2010. PMID: 20221443 Free PMC article.
-
Tetherin/BST-2: Restriction Factor or Immunomodulator?Curr HIV Res. 2016;14(3):235-46. doi: 10.2174/1570162x14999160224102752. Curr HIV Res. 2016. PMID: 26957198 Free PMC article. Review.
-
Tetherin and its viral antagonists.J Neuroimmune Pharmacol. 2011 Jun;6(2):188-201. doi: 10.1007/s11481-010-9256-1. Epub 2011 Jan 11. J Neuroimmune Pharmacol. 2011. PMID: 21222046 Free PMC article. Review.
Cited by
-
The RING finger protein family in health and disease.Signal Transduct Target Ther. 2022 Aug 30;7(1):300. doi: 10.1038/s41392-022-01152-2. Signal Transduct Target Ther. 2022. PMID: 36042206 Free PMC article. Review.
-
Mutations accumulated in the Spike of SARS-CoV-2 Omicron allow for more efficient counteraction of the restriction factor BST2/Tetherin.PLoS Pathog. 2024 Jan 8;20(1):e1011912. doi: 10.1371/journal.ppat.1011912. eCollection 2024 Jan. PLoS Pathog. 2024. PMID: 38190411 Free PMC article.
-
Beyond Tethering the Viral Particles: Immunomodulatory Functions of Tetherin (BST-2).DNA Cell Biol. 2019 Nov;38(11):1170-1177. doi: 10.1089/dna.2019.4777. Epub 2019 Sep 9. DNA Cell Biol. 2019. PMID: 31502877 Free PMC article. Review.
-
Tetherin restricts productive HIV-1 cell-to-cell transmission.PLoS Pathog. 2010 Jun 17;6(6):e1000955. doi: 10.1371/journal.ppat.1000955. PLoS Pathog. 2010. PMID: 20585562 Free PMC article.
-
The tumour suppressor APC promotes HIV-1 assembly via interaction with Gag precursor protein.Nat Commun. 2017 Jan 30;8:14259. doi: 10.1038/ncomms14259. Nat Commun. 2017. PMID: 28134256 Free PMC article.
References
-
- Goff SP. Host factors exploited by retroviruses. Nat Rev Microbiol. 2007;5:253–263. - PubMed
-
- Freed EO. HIV-1 and the host cell: an intimate association. Trends Microbiol. 2004;12:170–177. - PubMed
-
- Malim MH, Emerman M. HIV-1 accessory proteins–ensuring viral survival in a hostile environment. Cell Host Microbe. 2008;3:388–398. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

