Monotherapy with methotrexate for primary central nervous lymphoma has single agent activity in the absence of radiotherapy: a single institution cohort
- PMID: 20020180
- PMCID: PMC2883931
- DOI: 10.1007/s11060-009-0090-3
Monotherapy with methotrexate for primary central nervous lymphoma has single agent activity in the absence of radiotherapy: a single institution cohort
Abstract
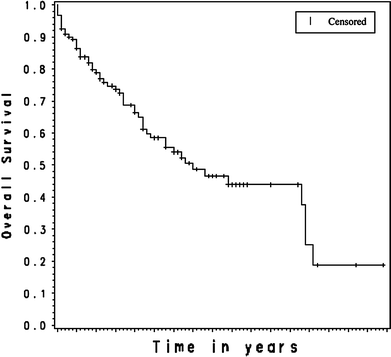
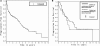
We have retrospectively reviewed toxicities and response of a cohort of primary central nervous system lymphoma (PCNSL) patients treated with high dose parenteral methotrexate (MTX) monotherapy without whole brain radiation. From The Massachusetts General Hospital (MGH) Cancer Registry, active since 1946, we selected all immunocompetent patients with histologic and/or radiographic PCNSL diagnosed between 1980 and 2007. We identified the recipients of MTX with leucovorin rescue as sole therapy. No patient received radiation therapy (XRT). We analyzed this cohort for toxicity, response and patterns of recurrence. The cohort of 121 patients received on average 11 cycles of intravenous MTX at a median dose of 8 g/m(2). Median interval between cycles was 10 days. After 3 months of therapy, the overall response rate was 85% (58% CR, 27% PR). The overall survival (OS) for the cohort was 7 years and progression-free survival (PFS) was 3.14 years. A trend toward a higher PFS was seen in patients who continued to receive MTX (3.48 years) every three months as compared to patients who ceased MTX after one year (2.86 years). Of 68 patients who achieved initial CR, there were 40 recurrences. Twenty-six of the 40 were re-induced with MTX as above; Sixty-nine percent again achieved CR. Eighty-one treatment-related toxicities occurred in 1316 MTX cycles. These toxicities included MRI white matter changes (N = 8) and lead to MTX cessation in 16 patients. High-dose MTX monotherapy of PCNSL is well-tolerated and provides PFS of >3 years and OS >7 years.
Figures
References
-
- Bradner WT. Pindell MH: myeloid leukemia C-1498 as a screen for cancer chemotherapeutic agents. Cancer Res. 1966;26:375–390. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

