Toll-like receptor 2-mediated peptidoglycan uptake by immature intestinal epithelial cells from apical side and exosome-associated transcellular transcytosis
- PMID: 20020500
- PMCID: PMC4414048
- DOI: 10.1002/jcp.21985
Toll-like receptor 2-mediated peptidoglycan uptake by immature intestinal epithelial cells from apical side and exosome-associated transcellular transcytosis
Abstract
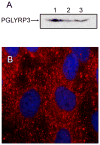
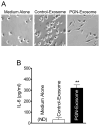
Peptidoglycan (PGN) is a potent immune adjuvant derived from bacterial cell walls. Previous investigations suggest that intestinal epithelium may absorb PGN from the lumen. Nonetheless, how PGN is taken up and crosses intestinal epithelium remains largely unclear. Here, we first characterized PGN transport in vitro using IEC-18 and HT29-CL19A cells, which represent less mature epithelial cells in intestinal crypts. With fluorescent microscopy, we visualized internalization of dual-labeled PGN by enterocytes. Engulfed PGN was found to form a complex with PGN recognition protein-3, which may facilitate delivering PGN in vivo. Utilizing electronic microscopy, we revealed that uptake of apical PGN across intestinal epithelial monolayers was involved in phagocytosis, multivesicular body formation, and exosome secretion. We also studied transport of PGN using the transwell system. Our data indicated that apically loaded PGN was exocytosed to the basolateral compartment with exosomes by HT29-CL19A cells. The PGN-contained basolateral exosome extracts induced macrophage activation. Through gavaging mice with labeled PGN, we found that luminal PGN was taken up by columnar epithelial cells in crypts of the small intestine. Furthermore, we showed that pre-confluent immature but not post-confluent mature C2BBe1 cells engulfed PGN via a toll-like receptor 2-dependent manner. Together, our findings suggest that (1) crypt-based immature intestinal epithelial cells play an important role in transport of luminal PGN over the intestinal epithelium; and (2) luminal PGN is transcytosed across intestinal epithelia via a toll-like receptor 2-mediated phagocytosis-multivesicular body-exosome pathway. The absorbed PGN and its derivatives may facilitate maintenance of intestinal immune homeostasis.
Conflict of interest statement
All authors declare that they have no conflict of interests to disclose.
Figures







Similar articles
-
Peptidoglycan recognition protein 3 (PglyRP3) has an anti-inflammatory role in intestinal epithelial cells.Immunobiology. 2012 Apr;217(4):412-9. doi: 10.1016/j.imbio.2011.10.013. Epub 2011 Nov 3. Immunobiology. 2012. PMID: 22099350
-
Mast cells play a crucial role in Staphylococcus aureus peptidoglycan-induced diarrhea.Am J Pathol. 2007 Aug;171(2):537-47. doi: 10.2353/ajpath.2007.061274. Epub 2007 Jun 28. Am J Pathol. 2007. PMID: 17600127 Free PMC article.
-
Bacterial peptidoglycan breaks down intestinal tolerance via mast cell activation: the role of TLR2 and NOD2.Immunol Cell Biol. 2007 Oct;85(7):538-45. doi: 10.1038/sj.icb.7100079. Epub 2007 Jun 12. Immunol Cell Biol. 2007. PMID: 17563761
-
Development, validation and implementation of an in vitro model for the study of metabolic and immune function in normal and inflamed human colonic epithelium.Dan Med J. 2015 Jan;62(1):B4973. Dan Med J. 2015. PMID: 25557335 Review.
-
Phenotypic and functional characterization of intestinal epithelial exosomes.Blood Cells Mol Dis. 2005 Jul-Aug;35(1):11-6. doi: 10.1016/j.bcmd.2005.04.001. Blood Cells Mol Dis. 2005. PMID: 15893486 Review.
Cited by
-
Outer membrane vesicles from Bacteroides fragilis contain coding and non-coding small RNA species that modulate inflammatory signaling in intestinal epithelial cells.bioRxiv [Preprint]. 2025 Jun 27:2025.06.25.661399. doi: 10.1101/2025.06.25.661399. bioRxiv. 2025. PMID: 40666935 Free PMC article. Preprint.
-
Molecular and functional profiling of apical versus basolateral small extracellular vesicles derived from primary human proximal tubular epithelial cells under inflammatory conditions.J Extracell Vesicles. 2021 Feb;10(4):e12064. doi: 10.1002/jev2.12064. Epub 2021 Feb 16. J Extracell Vesicles. 2021. PMID: 33643548 Free PMC article.
-
Production and characterization of avian crypt-villus enteroids and the effect of chemicals.BMC Vet Res. 2020 Jun 5;16(1):179. doi: 10.1186/s12917-020-02397-1. BMC Vet Res. 2020. PMID: 32503669 Free PMC article.
-
Luminal-applied flagellin is internalized by polarized intestinal epithelial cells and elicits immune responses via the TLR5 dependent mechanism.PLoS One. 2011;6(9):e24869. doi: 10.1371/journal.pone.0024869. Epub 2011 Sep 15. PLoS One. 2011. PMID: 21949773 Free PMC article.
-
The uptake of soluble and particulate antigens by epithelial cells in the mouse small intestine.PLoS One. 2014 Jan 27;9(1):e86656. doi: 10.1371/journal.pone.0086656. eCollection 2014. PLoS One. 2014. PMID: 24475164 Free PMC article.
References
-
- Augeron C, Laboisse CL. Emergence of permanently differentiated cell clones in a human colonic cancer cell line in culture after treatment with sodium butyrate. Cancer Res. 1984;44:3961–3969. - PubMed
-
- Boneca IG. The role of peptidoglycan in pathogenesis. Curr Opin Microbiol. 2005;8:46–53. - PubMed
-
- Bu HF, Wang X, Zhu YQ, Williams RY, Hsueh W, Zheng X, Rozenfeld RA, Zuo XL, Tan XD. Lysozyme-modified probiotic components protect rats against polymicrobial sepsis: role of macrophages and cathelicidin-related innate immunity. J Immunol. 2006;177:8767–8776. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

