Cellular injury and neuroinflammation in children with chronic intractable epilepsy
- PMID: 20021679
- PMCID: PMC2811703
- DOI: 10.1186/1742-2094-6-38
Cellular injury and neuroinflammation in children with chronic intractable epilepsy
Abstract
Objective: To elucidate the presence and potential involvement of brain inflammation and cell death in neurological morbidity and intractable seizures in childhood epilepsy, we quantified cell death, astrocyte proliferation, microglial activation and cytokine release in brain tissue from patients who underwent epilepsy surgery.
Methods: Cortical tissue was collected from thirteen patients with intractable epilepsy due to focal cortical dysplasia (6), encephalomalacia (5), Rasmussen's encephalitis (1) or mesial temporal lobe epilepsy (1). Sections were processed for immunohistochemistry using markers for neuron, astrocyte, microglia or cellular injury. Cytokine assay was performed on frozen cortices. Controls were autopsy brains from eight patients without history of neurological diseases.
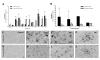
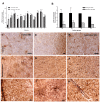
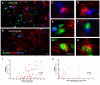
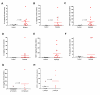
Results: Marked activation of microglia and astrocytes and diffuse cell death were observed in epileptogenic tissue. Numerous fibrillary astrocytes and their processes covered the entire cortex and converged on to blood vessels, neurons and microglia. An overwhelming number of neurons and astrocytes showed DNA fragmentation and its magnitude significantly correlated with seizure frequency. Majority of our patients with abundant cell death in the cortex have mental retardation. IL-1beta, IL-8, IL-12p70 and MIP-1beta were significantly increased in the epileptogenic cortex; IL-6 and MCP-1 were significantly higher in patients with family history of epilepsy.
Conclusions: Our results suggest that active neuroinflammation and marked cellular injury occur in pediatric epilepsy and may play a common pathogenic role or consequences in childhood epilepsy of diverse etiologies. Our findings support the concept that immunomodulation targeting activated microglia and astrocytes may be a novel therapeutic strategy to reduce neurological morbidity and prevent intractable epilepsy.
Figures

References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

