The tobacco MAP215/Dis1-family protein TMBP200 is required for the functional organization of microtubule arrays during male germline establishment
- PMID: 20022922
- PMCID: PMC2826647
- DOI: 10.1093/jxb/erp367
The tobacco MAP215/Dis1-family protein TMBP200 is required for the functional organization of microtubule arrays during male germline establishment
Abstract
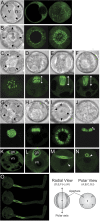
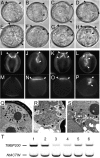
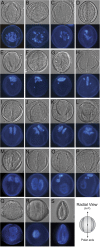
The haploid microspore division during pollen development in flowering plants is an intrinsically asymmetric division which establishes the male germline for sexual reproduction. Arabidopsis gem1 mutants lack the male germline as a result of disturbed microspore polarity, division asymmetry, and cytokinesis and represent loss-of-function mutants in MOR1/GEM1, a plant orthologue of the conserved MAP215/Dis1 microtubule associated protein (MAP) family. This provides genetic evidence for the role of MAP215/Dis1 in the organization of gametophytic microtubule arrays, but it has remained unknown how microtubule arrays are affected in gem1 mutant microspores. Here, novel male gametophytic microtubule-reporter Nicotiana tabacum plants were constructed, expressing a green fluorescent protein-alpha-TUBULIN fusion protein (GFP-TUA6) under the control of a microspore-specific promoter. These plants allow effective visualization of all major male gametophytic microtubule arrays and provide useful tools to study the regulation of microtubule arrays by MAPs and other effectors. Depletion of TMBP200, a tobacco homologue of MOR1/GEM1 in gametophytic microtubule-reporter plants using microspore-targeted RNA interference, induced defects in microspore polarity, division asymmetry and cytokinesis that were associated with striking defects in phragmoplast position, orientation, and structure. Our observations further reveal a requirement for TMBP200 in gametophytic spindle organization and a novel role in spindle position and orientation in polarized microspores. These results provide direct evidence for the function of MAP215/Dis1 family protein TMBP200 in the organization of microtubule arrays critical for male germline formation in plants.
Figures

Similar articles
-
A ticket for the live show: microtubules in male gametophyte development.Plant Signal Behav. 2010 May;5(5):614-7. doi: 10.4161/psb.11505. Epub 2010 Apr 20. Plant Signal Behav. 2010. PMID: 20404559 Free PMC article.
-
MAPping the eukaryotic tree of life: structure, function, and evolution of the MAP215/Dis1 family of microtubule-associated proteins.Int Rev Cytol. 2004;239:179-272. doi: 10.1016/S0074-7696(04)39004-2. Int Rev Cytol. 2004. PMID: 15464854 Review.
-
TMBP200, a XMAP215 homologue of tobacco BY-2 cells, has an essential role in plant mitosis.Protoplasma. 2011 Jul;248(3):493-502. doi: 10.1007/s00709-010-0189-6. Epub 2010 Aug 12. Protoplasma. 2011. PMID: 20703504
-
MOR1/GEM1 has an essential role in the plant-specific cytokinetic phragmoplast.Nat Cell Biol. 2002 Sep;4(9):711-4. doi: 10.1038/ncb844. Nat Cell Biol. 2002. PMID: 12198497 Free PMC article.
-
Microtubule organization and microtubule-associated proteins in plant cells.Int Rev Cell Mol Biol. 2014;312:1-52. doi: 10.1016/B978-0-12-800178-3.00001-4. Int Rev Cell Mol Biol. 2014. PMID: 25262237 Review.
Cited by
-
Effects of the protein GCP4 on gametophyte development in Arabidopsis thaliana.Protoplasma. 2021 May;258(3):483-493. doi: 10.1007/s00709-020-01520-1. Epub 2020 Nov 6. Protoplasma. 2021. PMID: 33155064
-
How plants LINC the SUN to KASH.Nucleus. 2013 May-Jun;4(3):206-15. doi: 10.4161/nucl.24088. Epub 2013 May 13. Nucleus. 2013. PMID: 23680964 Free PMC article. Review.
-
Regulation of Female Germline Specification via Small RNA Mobility in Arabidopsis.Plant Cell. 2020 Sep;32(9):2842-2854. doi: 10.1105/tpc.20.00126. Epub 2020 Jul 23. Plant Cell. 2020. PMID: 32703817 Free PMC article.
-
Using maize as a model to study pollen tube growth and guidance, cross-incompatibility and sperm delivery in grasses.Ann Bot. 2011 Sep;108(4):727-37. doi: 10.1093/aob/mcr017. Epub 2011 Feb 23. Ann Bot. 2011. PMID: 21345919 Free PMC article. Review.
-
Arabidopsis microtubule-associated protein MAP65-3 cross-links antiparallel microtubules toward their plus ends in the phragmoplast via its distinct C-terminal microtubule binding domain.Plant Cell. 2012 May;24(5):2071-85. doi: 10.1105/tpc.111.092569. Epub 2012 May 8. Plant Cell. 2012. PMID: 22570443 Free PMC article.
References
-
- Abe T, Hashimoto T. Altered microtubule dynamics by expression of modified α-tubulin protein causes right-handed helical growth in transgenic Arabidopsis plants. The Plant Journal. 2005;43:191–204. - PubMed
-
- Borg M, Brownfield L, Twell D. Male gametophyte development: a molecular perspective. Journal of Experimental Botany. 2009;60:1465–1478. - PubMed
-
- Burk DH, Zhong R, Morrison WH, III, Ye ZH. Disruption of cortical microtubules by overexpression of green fluorescent protein-tagged α-tubulin 6 causes a marked reduction in cell wall synthesis. Journal of Integrative Plant Biology. 2006;48:85–98.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

