Distinct subcellular localization of a group of synaptobrevin-like SNAREs in Paramecium tetraurelia and effects of silencing SNARE-specific chaperone NSF
- PMID: 20023070
- PMCID: PMC2823002
- DOI: 10.1128/EC.00220-09
Distinct subcellular localization of a group of synaptobrevin-like SNAREs in Paramecium tetraurelia and effects of silencing SNARE-specific chaperone NSF
Abstract
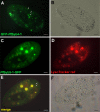
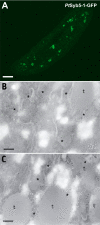
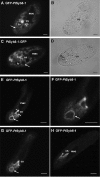
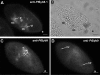
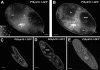
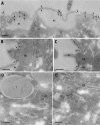
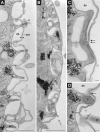
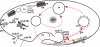
We have identified new synaptobrevin-like SNAREs and localized the corresponding gene products with green fluorescent protein (GFP)-fusion constructs and specific antibodies at the light and electron microscope (EM) levels. These SNAREs, named Paramecium tetraurelia synaptobrevins 8 to 12 (PtSyb8 to PtSyb12), showed mostly very restricted, specific localization, as they were found predominantly on structures involved in endo- or phagocytosis. In summary, we found PtSyb8 and PtSyb9 associated with the nascent food vacuole, PtSyb10 near the cell surface, at the cytostome, and in close association with ciliary basal bodies, and PtSyb11 on early endosomes and on one side of the cytostome, while PtSyb12 was found in the cytosol. PtSyb4 and PtSyb5 (identified previously) were localized on small vesicles, PtSyb5 probably being engaged in trichocyst (dense core secretory vesicle) processing. PtSyb4 and PtSyb5 are related to each other and are the furthest deviating of all SNAREs identified so far. Because they show no similarity with any other R-SNAREs outside ciliates, they may represent a ciliate-specific adaptation. PtSyb10 forms small domains near ciliary bases, and silencing slows down cell rotation during depolarization-induced ciliary reversal. NSF silencing supports a function of cell surface SNAREs by revealing vesicles along the cell membrane at sites normally devoid of vesicles. The distinct distributions of these SNAREs emphasize the considerable differentiation of membrane trafficking, particularly along the endo-/phagocytic pathway, in this protozoan.
Figures






References
-
- Adoutte A., Delgado P., Fleury A., Levilliers N., Laine M. C., Marty M. C., Boisvieux-Ulrich E., Sandoz D. 1991. Microtubule diversity in ciliated cells: evidence for its generation by post-translational modification in the axonemes of Paramecium and quail oviduct cells. Biol. Cell 71:227–245 - PubMed
-
- Allen R. D. 1984. Paramecium phagosome membrane: from oral region to cytoproct and back again. J. Protozool. 31:1–6
-
- Allen R. D., Fok A. K. 1993. Endosomal membrane traffic of ciliates, p. 57–83InPlattner H. (ed.), Advances in cell and molecular biology of membranes JAI Press, Greenwich, CT
-
- Allen R. D., Fok A. K. 2000. Membrane trafficking and processing in Paramecium. Int. Rev. Cytol. 198:277–318 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

