Role of protein tyrosine phosphatase SHP2 in barrier function of pulmonary endothelium
- PMID: 20023173
- PMCID: PMC2838678
- DOI: 10.1152/ajplung.00374.2009
Role of protein tyrosine phosphatase SHP2 in barrier function of pulmonary endothelium
Abstract
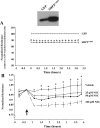
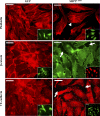
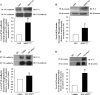
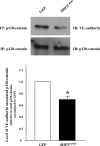
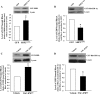
Pulmonary edema is mediated in part by disruption of interendothelial cell contacts. Protein tyrosine phosphatases (PTP) have been shown to affect both cell-extracellular matrix and cell-cell junctions. The SH2 domain-containing nonreceptor PTP, SHP2, is involved in intercellular signaling through direct interaction with adherens junction proteins. In this study, we examined the role of SHP2 in pulmonary endothelial barrier function. Inhibition of SHP2 promoted edema formation in rat lungs and increased monolayer permeability in cultured lung endothelial cells. In addition, pulmonary endothelial cells demonstrated a decreased level of p190RhoGAP activity following inhibition of SHP2, events that were accompanied by a concomitant increase in RhoA activity. Furthermore, immunofluorescence microscopy confirmed enhanced actin stress fiber formation and diminished interendothelial staining of adherens junction complex-associated proteins upon SHP2 inhibition. Finally, immunoprecipitation and immunoblot analyses demonstrated increased tyrosine phosphorylation of VE-cadherin, beta-catenin, and p190RhoGAP proteins, as well as decreased association between p120-catenin and VE-cadherin proteins. Our findings suggest that SHP2 supports basal pulmonary endothelial barrier function by coordinating the tyrosine phosphorylation profile of VE-cadherin, beta-catenin, and p190RhoGAP and the activity of RhoA, signaling molecules important in adherens junction complex integrity.
Figures
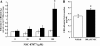

Similar articles
-
The tyrosine phosphatase SHP2 regulates recovery of endothelial adherens junctions through control of β-catenin phosphorylation.Mol Biol Cell. 2012 Nov;23(21):4212-25. doi: 10.1091/mbc.E12-01-0038. Epub 2012 Sep 5. Mol Biol Cell. 2012. PMID: 22956765 Free PMC article.
-
Fibroblast growth factor signaling potentiates VE-cadherin stability at adherens junctions by regulating SHP2.PLoS One. 2012;7(5):e37600. doi: 10.1371/journal.pone.0037600. Epub 2012 May 22. PLoS One. 2012. PMID: 22629427 Free PMC article.
-
SH2 domain-containing protein tyrosine phosphatase 2 and focal adhesion kinase protein interactions regulate pulmonary endothelium barrier function.Am J Respir Cell Mol Biol. 2015 Jun;52(6):695-707. doi: 10.1165/rcmb.2013-0489OC. Am J Respir Cell Mol Biol. 2015. PMID: 25317600 Free PMC article.
-
Dynamics between actin and the VE-cadherin/catenin complex: novel aspects of the ARP2/3 complex in regulation of endothelial junctions.Cell Adh Migr. 2014;8(2):125-35. doi: 10.4161/cam.28243. Cell Adh Migr. 2014. PMID: 24621569 Free PMC article. Review.
-
Dynamic Regulation of Vascular Permeability by Vascular Endothelial Cadherin-Mediated Endothelial Cell-Cell Junctions.J Nippon Med Sch. 2017;84(4):148-159. doi: 10.1272/jnms.84.148. J Nippon Med Sch. 2017. PMID: 28978894 Review.
Cited by
-
KRIT1: A Traffic Warden at the Busy Crossroads Between Redox Signaling and the Pathogenesis of Cerebral Cavernous Malformation Disease.Antioxid Redox Signal. 2023 Mar;38(7-9):496-528. doi: 10.1089/ars.2021.0263. Epub 2022 Nov 1. Antioxid Redox Signal. 2023. PMID: 36047808 Free PMC article. Review.
-
Novel regulators of endothelial barrier function.Am J Physiol Lung Cell Mol Physiol. 2014 Dec 15;307(12):L924-35. doi: 10.1152/ajplung.00318.2014. Epub 2014 Nov 7. Am J Physiol Lung Cell Mol Physiol. 2014. PMID: 25381026 Free PMC article. Review.
-
Interleukin 2 Activates Brain Microvascular Endothelial Cells Resulting in Destabilization of Adherens Junctions.J Biol Chem. 2016 Oct 28;291(44):22913-22923. doi: 10.1074/jbc.M116.729038. Epub 2016 Sep 6. J Biol Chem. 2016. PMID: 27601468 Free PMC article.
-
ZINC40099027 activates human focal adhesion kinase by accelerating the enzymatic activity of the FAK kinase domain.Pharmacol Res Perspect. 2021 Apr;9(2):e00737. doi: 10.1002/prp2.737. Pharmacol Res Perspect. 2021. PMID: 33715263 Free PMC article.
-
The tyrosine phosphatase SHP2 regulates recovery of endothelial adherens junctions through control of β-catenin phosphorylation.Mol Biol Cell. 2012 Nov;23(21):4212-25. doi: 10.1091/mbc.E12-01-0038. Epub 2012 Sep 5. Mol Biol Cell. 2012. PMID: 22956765 Free PMC article.
References
-
- Andriopoulou P, Navarro P, Zanetti A, Lampugnani MG, Dejana E. Histamine induces tyrosine phosphorylation of endothelial cell-to-cell adherens junctions. Arterioscler Thromb Vasc Biol 19: 2286–2297, 1999 - PubMed
-
- Angelini DJ, Hyun SW, Grigoryev DN, Garg P, Gong P, Singh IS, Passaniti A, Hasday JD, Goldblum SE. TNF-α increases tyrosine phosphorylation of vascular endothelial cadherin and opens the paracellular pathway through fyn activation in human lung endothelia. Am J Physiol Lung Cell Mol Physiol 291: L1232–L1245, 2006 - PubMed
-
- Arthur WT, Petch LA, Burridge K. Integrin engagement suppresses RhoA activity via a c-Src-dependent mechanism. Curr Biol 10: 719–722, 2000 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

