Functional importance of the NH2-terminal insertion sequence of lung surfactant protein B
- PMID: 20023175
- PMCID: PMC2838675
- DOI: 10.1152/ajplung.00190.2009
Functional importance of the NH2-terminal insertion sequence of lung surfactant protein B
Abstract
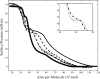
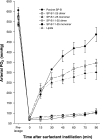
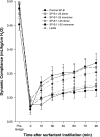
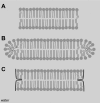
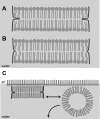
Lung surfactant protein B (SP-B) is required for proper surface activity of pulmonary surfactant. In model lung surfactant lipid systems composed of saturated and unsaturated lipids, the unsaturated lipids are removed from the film at high compression. It is thought that SP-B helps anchor these lipids closely to the monolayer in three-dimensional cylindrical structures termed "nanosilos" seen by atomic force microscopy imaging of deposited monolayers at high surface pressures. Here we explore the role of the SP-B NH(2) terminus in the formation and stability of these cylindrical structures, specifically the distribution of lipid stack height, width, and density with four SP-B truncation peptides: SP-B 1-25, SP-B 9-25, SP-B 11-25, and SP-B 1-25Nflex (prolines 2 and 4 substituted with alanine). The first nine amino acids, termed the insertion sequence and the interface seeking tryptophan residue 9, are shown to stabilize the formation of nanosilos while an increase in the insertion sequence flexibility (SP-B 1-25Nflex) may improve peptide functionality. This provides a functional understanding of the insertion sequence beyond anchoring the protein to the two-dimensional membrane lining the lung, as it also stabilizes formation of nanosilos, creating reversible repositories for fluid lipids at high compression. In lavaged, surfactant-deficient rats, instillation of a mixture of SP-B 1-25 (as a monomer or dimer) and synthetic lung lavage lipids quickly improved oxygenation and dynamic compliance, whereas SP-B 11-25 surfactants showed oxygenation and dynamic compliance values similar to that of lipids alone, demonstrating a positive correlation between formation of stable, but reversible, nanosilos and in vivo efficacy.
Figures



Similar articles
-
Critical structure-function determinants within the N-terminal region of pulmonary surfactant protein SP-B.Biophys J. 2006 Jan 1;90(1):238-49. doi: 10.1529/biophysj.105.073403. Epub 2005 Oct 7. Biophys J. 2006. PMID: 16214863 Free PMC article.
-
Kinetics of phospholipid insertion into monolayers containing the lung surfactant proteins SP-B or SP-C.Eur Biophys J. 2002 Mar;31(1):52-61. doi: 10.1007/s002490100181. Eur Biophys J. 2002. PMID: 12046897
-
Surfactant with SP-B and SP-C analogues improves lung function in surfactant-deficient rats.Biol Neonate. 2002;82(3):181-7. doi: 10.1159/000063607. Biol Neonate. 2002. PMID: 12373069
-
Protein-lipid interactions and surface activity in the pulmonary surfactant system.Chem Phys Lipids. 2006 Jun;141(1-2):105-18. doi: 10.1016/j.chemphyslip.2006.02.017. Epub 2006 Mar 20. Chem Phys Lipids. 2006. PMID: 16600200 Review.
-
Hydrophobic surfactant proteins and their analogues.Neonatology. 2007;91(4):303-10. doi: 10.1159/000101346. Epub 2007 Jun 7. Neonatology. 2007. PMID: 17575474 Review.
Cited by
-
Structure-function correlations of pulmonary surfactant protein SP-B and the saposin-like family of proteins.Eur Biophys J. 2013 Mar;42(2-3):209-22. doi: 10.1007/s00249-012-0858-9. Epub 2012 Sep 21. Eur Biophys J. 2013. PMID: 22996193 Review.
-
Lung surfactant protein SP-B promotes formation of bilayer reservoirs from monolayer and lipid transfer between the interface and subphase.Biophys J. 2011 Apr 6;100(7):1678-87. doi: 10.1016/j.bpj.2011.02.019. Biophys J. 2011. PMID: 21463581 Free PMC article.
-
Surfactant protein C peptides with salt-bridges ("ion-locks") promote high surfactant activities by mimicking the α-helix and membrane topography of the native protein.PeerJ. 2014 Jul 15;2:e485. doi: 10.7717/peerj.485. eCollection 2014. PeerJ. 2014. PMID: 25083348 Free PMC article.
-
All-Atom Molecular Dynamics Simulations of Dimeric Lung Surfactant Protein B in Lipid Multilayers.Int J Mol Sci. 2019 Aug 8;20(16):3863. doi: 10.3390/ijms20163863. Int J Mol Sci. 2019. PMID: 31398818 Free PMC article.
-
Design of Surfactant Protein B Peptide Mimics Based on the Saposin Fold for Synthetic Lung Surfactants.Biomed Hub. 2016 Sep-Dec;1(3):1-21. doi: 10.1159/000451076. Epub 2016 Nov 16. Biomed Hub. 2016. PMID: 28503550 Free PMC article.
References
-
- Anzueto A, Baughman RP, Guntupalli KK, Weg JG, Weidemann HP, Raventos AA, Lemaire F, Long W, Zaccardelli DS, Pattishall EN. Aerosolized surfactant in adults with sepsis-induced acute respiratory distress syndrome. N Engl J Med 334: 1417–1421, 1996 - PubMed
-
- Avery ME, Mead J. Surface properties in relation to atelectasis and hyaline membrane disease. Am J Dis Child 97: 517–523, 1959 - PubMed
-
- Baatz JE, Zou Y, Cox JT, Wang Z, Notter RH. High-yield purification of lung surfactant proteins SP-B and SP-C and the effects on surface activity. Protein Expr Purif 23: 180–190, 2001 - PubMed
-
- Berggren P, Curstedt T, Grossmann G, Nilsson R, Robertson B. Physiological activity of pulmonary surfactant with low protein content: effect of enrichment with synthetic phospholipids. Exp Lung Res 8: 29–51, 1985 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

