In vivo characterization of activatable cell penetrating peptides for targeting protease activity in cancer
- PMID: 20023745
- PMCID: PMC2796841
- DOI: 10.1039/b904890a
In vivo characterization of activatable cell penetrating peptides for targeting protease activity in cancer
Abstract
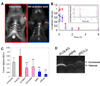
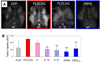
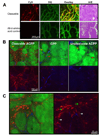
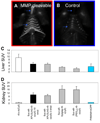
Activatable cell penetrating peptides (ACPPs) are novel in vivo targeting agents comprised of a polycationic cell penetrating peptide (CPP) connected via a cleavable linker to a neutralizing polyanion (). Adsorption and uptake into cells are inhibited until the linker is proteolyzed. An ACPP cleavable by matrix metalloproteinase-2 (MMP-2) in vitro was the first one demonstrated to work in a tumor model in vivo, but only HT-1080 xenografts and resected human squamous cell carcinomas were tested. Generality to other cancer types, in vivo selectivity of ACPPs for MMPs, and spatial resolution require further characterization. We now show that ACPPs can target many xenograft tumor models from different cancer sites, as well as a thoroughly studied transgenic model of spontaneous breast cancer (mouse mammary tumor virus promoter driving polyoma middle T antigen, MMTV-PyMT). Pharmacological inhibitors and genetic knockouts indicate that current ACPPs are selective for MMP-2 and MMP-9 in the above in vivo models. In accord with the known local distribution of MMP activity, accumulation is strongest at the tumor-stromal interface in primary tumors and associated metastases, indicating better spatial resolution (<50 mum) than other currently available MMP-cleavable probes. We also find that background uptake of ACPPs into normal tissues such as cartilage can be decreased by appending inert macromolecules of 30-50 KDa to the polyanionic inhibitory domain. Our results validate an approach that should generally deliver imaging agents and chemotherapeutics to sites of invasion, tumor-promoting inflammation, and metastasis.
Figures

Similar articles
-
Systemic in vivo distribution of activatable cell penetrating peptides is superior to that of cell penetrating peptides.Integr Biol (Camb). 2009 Jun;1(5-6):371-81. doi: 10.1039/b904878b. Epub 2009 May 11. Integr Biol (Camb). 2009. PMID: 20023744 Free PMC article.
-
Tumor targeting of MMP-2/9 activatable cell-penetrating imaging probes is caused by tumor-independent activation.J Nucl Med. 2011 Feb;52(2):279-86. doi: 10.2967/jnumed.110.082503. Epub 2011 Jan 13. J Nucl Med. 2011. PMID: 21233187
-
In vivo biodistribution of radiolabeled MMP-2/9 activatable cell-penetrating peptide probes in tumor-bearing mice.Contrast Media Mol Imaging. 2015 Jan-Feb;10(1):59-66. doi: 10.1002/cmmi.1605. Epub 2014 May 13. Contrast Media Mol Imaging. 2015. PMID: 24823643
-
Cy5.5-Conjugated matrix metalloproteinase cleavable peptide nanoprobe.2012 Mar 19 [updated 2012 Apr 12]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. 2012 Mar 19 [updated 2012 Apr 12]. In: Molecular Imaging and Contrast Agent Database (MICAD) [Internet]. Bethesda (MD): National Center for Biotechnology Information (US); 2004–2013. PMID: 22514808 Free Books & Documents. Review.
-
[The application of enzyme-sensitive activatable cell-penetrating peptides to targeted delivery system].Yao Xue Xue Bao. 2015 Feb;50(2):141-7. Yao Xue Xue Bao. 2015. PMID: 25975019 Review. Chinese.
Cited by
-
Length-dependent proteolytic cleavage of short oligopeptides catalyzed by matrix metalloprotease-9.Biopolymers. 2013 Nov;100(6):790-795. doi: 10.1002/bip.22240. Biopolymers. 2013. PMID: 23520037 Free PMC article.
-
Combined TP53 mutation/3p loss correlates with decreased radiosensitivity and increased matrix-metalloproteinase activity in head and neck carcinoma.Oral Oncol. 2015 May;51(5):470-5. doi: 10.1016/j.oraloncology.2015.01.014. Epub 2015 Feb 27. Oral Oncol. 2015. PMID: 25735654 Free PMC article.
-
Enhancing cellular uptake of activable cell-penetrating peptide-doxorubicin conjugate by enzymatic cleavage.Int J Nanomedicine. 2012;7:1613-21. doi: 10.2147/IJN.S30104. Epub 2012 Mar 26. Int J Nanomedicine. 2012. PMID: 22619516 Free PMC article.
-
Impact of MMP-2 and MMP-9 enzyme activity on wound healing, tumor growth and RACPP cleavage.PLoS One. 2018 Sep 24;13(9):e0198464. doi: 10.1371/journal.pone.0198464. eCollection 2018. PLoS One. 2018. PMID: 30248101 Free PMC article.
-
Molecular imaging for cancer diagnosis and surgery.Adv Drug Deliv Rev. 2014 Feb;66:90-100. doi: 10.1016/j.addr.2013.09.007. Epub 2013 Sep 22. Adv Drug Deliv Rev. 2014. PMID: 24064465 Free PMC article. Review.
References
-
- Goun EA, et al. Intracellular Cargo Delivery by an Octaarginine Transporter Adapted to Target Prostate Cancer Cells through Cell Surface Protease Activation. Bioconjugate Chem. 2006;17:787–796. - PubMed
-
- Bremer C, Bredow S, Mahmood U, Weissleder R, Tung CH. Optical imaging of matrix metalloproteinase-2 activity in tumors: feasibility study in a mouse model. Radiology. 2001;221:523–529. - PubMed
-
- Chabottaux V, Noel A. Breast cancer progression: insights into multifaceted matrix metalloproteinases. Clin Exp Metastasis. 2007;24:647–656. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

