Mutation of conserved cysteines in the Ly6 domain of GPIHBP1 in familial chylomicronemia
- PMID: 20026666
- PMCID: PMC3035517
- DOI: 10.1194/jlr.M002717
Mutation of conserved cysteines in the Ly6 domain of GPIHBP1 in familial chylomicronemia
Abstract
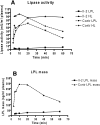
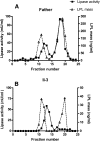
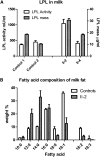
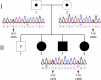
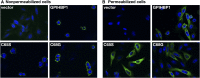
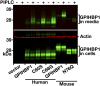
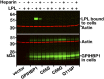
We investigated a family from northern Sweden in which three of four siblings have congenital chylomicronemia. LPL activity and mass in pre- and postheparin plasma were low, and LPL release into plasma after heparin injection was delayed. LPL activity and mass in adipose tissue biopsies appeared normal. [(35)S]Methionine incorporation studies on adipose tissue showed that newly synthesized LPL was normal in size and normally glycosylated. Breast milk from the affected female subjects contained normal to elevated LPL mass and activity levels. The milk had a lower than normal milk lipid content, and the fatty acid composition was compatible with the milk lipids being derived from de novo lipogenesis, rather than from the plasma lipoproteins. Given the delayed release of LPL into the plasma after heparin, we suspected that the chylomicronemia might be caused by mutations in GPIHBP1. Indeed, all three affected siblings were compound heterozygotes for missense mutations involving highly conserved cysteines in the Ly6 domain of GPIHBP1 (C65S and C68G). The mutant GPIHBP1 proteins reached the surface of transfected Chinese hamster ovary cells but were defective in their ability to bind LPL (as judged by both cell-based and cell-free LPL binding assays). Thus, the conserved cysteines in the Ly6 domain are crucial for GPIHBP1 function.
Figures
References
-
- Wang H., Eckel R. H. 2009. Lipoprotein lipase: from gene to obesity. Am. J. Physiol. Endocrinol. Metab. 297: E271–E288. - PubMed
-
- Olivecrona T., Olivecrona G. 2009. The ins and outs of adipose tissue. In Cellular Lipid Mmetabolism Ehnholm C., editor Springer, Heidelberg, Germany: 315–369.
-
- Krieger M., Herz J. 1994. Structures and functions of multiligand lipoprotein receptors: macrophage scavenger receptors and LDL receptor-related protein (LRP). Annu. Rev. Biochem. 63: 601–637. - PubMed
-
- Beisiegel U. 1995. Receptors for triglyceride-rich lipoproteins and their role in lipoprotein metabolism. Curr. Opin. Lipidol. 6: 117–122. - PubMed
-
- Gliemann J. 1998. Receptors of the low density lipoprotein (LDL) receptor family in man. Multiple functions of the large family members via interaction with complex ligands. Biol. Chem. 379: 951–964. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

