Fatty acid synthase impacts the pathobiology of Candida parapsilosis in vitro and during mammalian infection
- PMID: 20027295
- PMCID: PMC2791871
- DOI: 10.1371/journal.pone.0008421
Fatty acid synthase impacts the pathobiology of Candida parapsilosis in vitro and during mammalian infection
Abstract
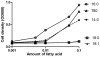
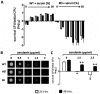
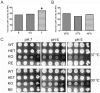
Cytosolic fungal fatty acid synthase is composed of two subunits alpha and beta, which are encoded by Fas1 and Fas2 genes. In this study, the Fas2 genes of the human pathogen Candida parapsilosis were deleted using a modified SAT1 flipper technique. CpFas2 was essential in media lacking exogenous fatty acids and the growth of Fas2 disruptants (Fas2 KO) was regulated by the supplementation of different long chain fatty acids, such as myristic acid (14ratio0), palmitic acid (16ratio0), and Tween 80, in a dose-specific manner. Lipidomic analysis revealed that Fas2 KO cells were severely restricted in production of unsaturated fatty acids. The Fas2 KO strains were unable to form normal biofilms and were more efficiently killed by murine-like macrophages, J774.16, than the wild type, heterozygous and reconstituted strains. Furthermore, Fas2 KO yeast were significantly less virulent in a systemic murine infection model. The Fas2 KO cells were also hypersensitive to human serum, and inhibition of CpFas2 in WT C. parapsilosis by cerulenin significantly decreased fungal growth in human serum. This study demonstrates that CpFas2 is essential for C. parapsilosis growth in the absence of exogenous fatty acids, is involved in unsaturated fatty acid production, influences fungal virulence, and represents a promising antifungal drug target.
Conflict of interest statement
Figures

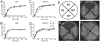



Similar articles
-
Inhibition of Candida parapsilosis fatty acid synthase (Fas2) induces mitochondrial cell death in serum.PLoS Pathog. 2012;8(8):e1002879. doi: 10.1371/journal.ppat.1002879. Epub 2012 Aug 30. PLoS Pathog. 2012. PMID: 22952445 Free PMC article.
-
The stearoyl-coenzyme A desaturase 1 is essential for virulence and membrane stress in Candida parapsilosis through unsaturated fatty acid production.Infect Immun. 2011 Jan;79(1):136-45. doi: 10.1128/IAI.00753-10. Epub 2010 Oct 25. Infect Immun. 2011. PMID: 20974817 Free PMC article.
-
Lipid droplet formation protects against gluco/lipotoxicity in Candida parapsilosis: an essential role of fatty acid desaturase Ole1.Cell Cycle. 2011 Sep 15;10(18):3159-67. doi: 10.4161/cc.10.18.16932. Epub 2011 Sep 15. Cell Cycle. 2011. PMID: 21897120
-
Candida parapsilosis Secreted Lipase as an Important Virulence Factor.Curr Protein Pept Sci. 2017;18(10):1043-1049. doi: 10.2174/1389203717666160813163054. Curr Protein Pept Sci. 2017. PMID: 27526931 Review.
-
Candida and candidaemia. Susceptibility and epidemiology.Dan Med J. 2013 Nov;60(11):B4698. Dan Med J. 2013. PMID: 24192246 Review.
Cited by
-
Phenotypic and Genotypic Characterization of Candida parapsilosis complex isolates from a Lebanese Hospital.Res Sq [Preprint]. 2024 May 30:rs.3.rs-4169036. doi: 10.21203/rs.3.rs-4169036/v1. Res Sq. 2024. Update in: Sci Rep. 2025 Feb 10;15(1):4853. doi: 10.1038/s41598-024-84535-5. PMID: 38903078 Free PMC article. Updated. Preprint.
-
The Trisubstituted Isoxazole MMV688766 Exerts Broad-Spectrum Activity against Drug-Resistant Fungal Pathogens through Inhibition of Lipid Homeostasis.mBio. 2022 Dec 20;13(6):e0273022. doi: 10.1128/mbio.02730-22. Epub 2022 Oct 27. mBio. 2022. PMID: 36300931 Free PMC article.
-
Inhibition of cell cycle-dependent hyphal and biofilm formation by a novel cytochalasin 19,20‑epoxycytochalasin Q in Candida albicans.Sci Rep. 2023 Jun 15;13(1):9724. doi: 10.1038/s41598-023-36191-4. Sci Rep. 2023. PMID: 37322086 Free PMC article.
-
Integrative functional analysis uncovers metabolic differences between Candida species.Commun Biol. 2022 Sep 26;5(1):1013. doi: 10.1038/s42003-022-03955-z. Commun Biol. 2022. PMID: 36163459 Free PMC article.
-
Candida parapsilosis and the neonate: epidemiology, virulence and host defense in a unique patient setting.Expert Rev Anti Infect Ther. 2012 Aug;10(8):935-46. doi: 10.1586/eri.12.74. Expert Rev Anti Infect Ther. 2012. PMID: 23030332 Free PMC article. Review.
References
-
- Martin GS, Mannino DM, Eaton S, Moss M. The epidemiology of sepsis in the United States from 1979 through 2000. N Engl J Med. 2003;348:1546–1554. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

