Dynamic mechanical loading enhances functional properties of tissue-engineered cartilage using mature canine chondrocytes
- PMID: 20028219
- PMCID: PMC2952125
- DOI: 10.1089/ten.TEA.2009.0482
Dynamic mechanical loading enhances functional properties of tissue-engineered cartilage using mature canine chondrocytes
Abstract
Objective: The concept of cartilage functional tissue engineering (FTE) has promoted the use of physiologic loading bioreactor systems to cultivate engineered tissues with load-bearing properties. Prior studies have demonstrated that culturing agarose constructs seeded with primary bovine chondrocytes from immature joints, and subjected to dynamic deformation, produced equilibrium compressive properties and proteoglycan content matching the native tissue. In the process of translating these results to an adult canine animal model, it was found that protocols previously successful with immature bovine primary chondrocytes did not produce the same successful outcome when using adult canine primary chondrocytes. The objective of this study was to assess the efficacy of a modified FTE protocol using adult canine chondrocytes seeded in agarose hydrogel and subjected to dynamic loading.
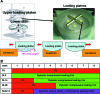
Method: Two modes of dynamic loading were applied to constructs using custom bioreactors: unconfined axial compressive deformational loading (DL; 1 Hz, 10% deformation) or sliding contact loading (Slide; 0.5 Hz, 10% deformation). Loading for 3 h daily was initiated on day 0, 14, or 28 (DL0, DL14, DL28, and Slide14).
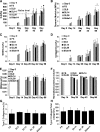
Results: Constructs with applied loading (both DL and Slide) exhibited significant increases in Young's modulus compared with free-swelling control as early as day 28 in culture (p < 0.05). However, glycosaminoglycan, collagen, and DNA content were not statistically different among the various groups. The modulus values attained for engineered constructs compare favorably with (and exceed in some cases) those of native canine knee (patella groove and condyle) cartilage.
Conclusion: Our findings successfully demonstrate an FTE strategy incorporating clinically relevant, adult chondrocytes and gel scaffold for engineering cartilage replacement tissue. These results, using continuous growth factor supplementation, are in contrast to our previously reported studies with immature chondrocytes where the sequential application of dynamic loading after transient transforming growth factor-beta3 application was found to be a superior culture protocol. Sliding, which simulates aspects of joint articulation, has shown promise in promoting engineered tissue development and provides an alternative option for FTE of cartilage constructs to be further explored.
Figures
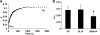


Similar articles
-
The beneficial effect of delayed compressive loading on tissue-engineered cartilage constructs cultured with TGF-beta3.Osteoarthritis Cartilage. 2007 Sep;15(9):1025-33. doi: 10.1016/j.joca.2007.03.008. Epub 2007 May 10. Osteoarthritis Cartilage. 2007. PMID: 17498976 Free PMC article.
-
The role of cell seeding density and nutrient supply for articular cartilage tissue engineering with deformational loading.Osteoarthritis Cartilage. 2003 Dec;11(12):879-90. doi: 10.1016/j.joca.2003.08.006. Osteoarthritis Cartilage. 2003. PMID: 14629964
-
A paradigm for functional tissue engineering of articular cartilage via applied physiologic deformational loading.Ann Biomed Eng. 2004 Jan;32(1):35-49. doi: 10.1023/b:abme.0000007789.99565.42. Ann Biomed Eng. 2004. PMID: 14964720 Review.
-
The effect of applied compressive loading on tissue-engineered cartilage constructs cultured with TGF-beta3.Conf Proc IEEE Eng Med Biol Soc. 2006;2006:779-82. doi: 10.1109/IEMBS.2006.259313. Conf Proc IEEE Eng Med Biol Soc. 2006. PMID: 17946858
-
Shape, loading, and motion in the bioengineering design, fabrication, and testing of personalized synovial joints.J Biomech. 2010 Jan 5;43(1):156-65. doi: 10.1016/j.jbiomech.2009.09.021. Epub 2009 Oct 7. J Biomech. 2010. PMID: 19815214 Free PMC article. Review.
Cited by
-
A Friction Testing-Bioreactor Device for Study of Synovial Joint Biomechanics, Mechanobiology, and Physical Regulation.J Vis Exp. 2022 Jun 2;(184):10.3791/63880. doi: 10.3791/63880. J Vis Exp. 2022. PMID: 35723474 Free PMC article.
-
Deciphering mechanical regulation of chondrogenesis in fibrin-polyurethane composite scaffolds enriched with human mesenchymal stem cells: a dual computational and experimental approach.Tissue Eng Part A. 2014 Apr;20(7-8):1197-212. doi: 10.1089/ten.TEA.2013.0145. Epub 2014 Jan 11. Tissue Eng Part A. 2014. PMID: 24199606 Free PMC article.
-
A novel bioreactor system for biaxial mechanical loading enhances the properties of tissue-engineered human cartilage.Sci Rep. 2017 Dec 5;7(1):16997. doi: 10.1038/s41598-017-16523-x. Sci Rep. 2017. PMID: 29208903 Free PMC article.
-
A Systems Approach to Biomechanics, Mechanobiology, and Biotransport.J Biomech Eng. 2024 Apr 1;146(4):040801. doi: 10.1115/1.4064547. J Biomech Eng. 2024. PMID: 38270930 Free PMC article.
-
The development of a mature collagen network in cartilage from human bone marrow stem cells in Transwell culture.Matrix Biol. 2016 Mar;50:16-26. doi: 10.1016/j.matbio.2015.10.003. Epub 2015 Oct 30. Matrix Biol. 2016. PMID: 26523516 Free PMC article.
References
-
- Caplan A.I. Elyaderani M. Mochizuki Y. Wakitani S. Goldberg V.M. Principles of cartilage repair and regeneration. Clin Orthop. 1997;342:254. - PubMed
-
- Caplan A.I. Goto T. Wakitani S. Pineda S.J. Haynesworth S.E. Goldbery V.M. Cell-based technologies for cartilage repair. In: Finerman G.A.M., editor; Noyes F.R., editor. Biology and Biomechanics of the Traumatized Synovial Joint: The Knee as a Model. Rosemont, IL: AAOS; 1993. pp. 111–122.
-
- Gao J. Dennis J.E. Solchaga L.A. Goldberg V.M. Caplan A.I. Repair of osteochondral defect with tissue-engineered two-phase composite material of injectable calcium phosphate and hyaluronan sponge. Tissue Eng. 2002;5:827. - PubMed
-
- Kawamura S. Wakitani S. Kimura T. Maeda A. Caplan A.I. Shino K. Ochi T. Articular cartilage repair. Rabbit experiments with a collagen gel-biomatrix and chondrocytes cultured in it. Acta Orthop Scand. 1998;69:56. - PubMed
-
- Orr T.E. Patel A.M. Wong B. Hatzigiannis G.P. Minas T. Spector M. Attachment of periosteal grafts to articular cartilage with fibrin sealent. J Biomed Mater Res. 1999;44:308. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

