A foreign protein incorporated on the Tip of T3 pili in Lactococcus lactis elicits systemic and mucosal immunity
- PMID: 20028807
- PMCID: PMC2825955
- DOI: 10.1128/IAI.01037-09
A foreign protein incorporated on the Tip of T3 pili in Lactococcus lactis elicits systemic and mucosal immunity
Abstract
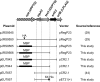
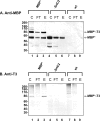
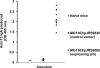
The use of Lactococcus lactis to deliver a chosen antigen to the mucosal surface has been shown to elicit an immune response in mice and is a possible method of vaccination in humans. The recent discovery on Gram-positive bacteria of pili that are covalently attached to the bacterial surface and the elucidation of the residues linking the major and minor subunits of such pili suggests that the presentation of an antigen on the tip of pili external to the surface of L. lactis might constitute a successful vaccine strategy. As a proof of principle, we have fused a foreign protein (the Escherichia coli maltose-binding protein) to the C-terminal region of the native tip protein (Cpa) of the T3 pilus derived from Streptococcus pyogenes and expressed this fusion protein (MBP*) in L. lactis. We find that MBP* is incorporated into pili in this foreign host, as shown by Western blot analyses of cell wall proteins and by immunogold electron microscopy. Furthermore, since the MBP* on these pili retains its native biological activity, it appears to retain its native structure. Mucosal immunization of mice with this L. lactis strain expressing pilus-linked MBP* results in production of both a systemic and a mucosal response (IgG and IgA antibodies) against the MBP antigen. We suggest that this type of mucosal vaccine delivery system, which we term UPTOP (for unhindered presentation on tips of pili), may provide an inexpensive and stable alternative to current mechanisms of immunization for many serious human pathogens.
Figures





Similar articles
-
Oral Immunization with a Recombinant Lactococcus lactis-Expressing HIV-1 Antigen on Group A Streptococcus Pilus Induces Strong Mucosal Immunity in the Gut.J Immunol. 2015 Nov 15;195(10):5025-34. doi: 10.4049/jimmunol.1501243. Epub 2015 Oct 19. J Immunol. 2015. PMID: 26482408 Free PMC article.
-
Mucosal and systemic immune responses to chimeric fimbriae expressed by Salmonella enterica serovar typhimurium vaccine strains.Infect Immun. 2000 Jun;68(6):3129-39. doi: 10.1128/IAI.68.6.3129-3139.2000. Infect Immun. 2000. PMID: 10816454 Free PMC article.
-
Heterologous expression of Intimin and IpaB fusion protein in Lactococcus lactis and its mucosal delivery elicit protection against pathogenicity of Escherichia coli O157 and Shigella flexneri in a murine model.Int Immunopharmacol. 2020 Aug;85:106617. doi: 10.1016/j.intimp.2020.106617. Epub 2020 May 25. Int Immunopharmacol. 2020. PMID: 32464569
-
Lactococcus lactis-based vaccines: current status and future perspectives.Hum Vaccin. 2011 Jan 1;7(1):106-9. doi: 10.4161/hv.7.1.13631. Epub 2011 Jan 1. Hum Vaccin. 2011. PMID: 21263226 Review.
-
[Progress on lactococcus lactis expressing heterologous antigens as live mucosal vaccines].Wei Sheng Wu Xue Bao. 2006 Aug;46(4):680-3. Wei Sheng Wu Xue Bao. 2006. PMID: 17037080 Review. Chinese.
Cited by
-
Neutralizing-antibody-mediated protection of chickens against infectious bursal disease via one-time vaccination with inactivated recombinant Lactococcus lactis expressing a fusion protein constructed from the RCK protein of Salmonella enterica and VP2 of infectious bursal disease virus.Microb Cell Fact. 2019 Jan 31;18(1):21. doi: 10.1186/s12934-019-1061-9. Microb Cell Fact. 2019. PMID: 30704494 Free PMC article.
-
PilVax - a novel peptide delivery platform for the development of mucosal vaccines.Sci Rep. 2018 Feb 7;8(1):2555. doi: 10.1038/s41598-018-20863-7. Sci Rep. 2018. PMID: 29416095 Free PMC article.
-
Functional identification of conserved residues involved in Lactobacillus rhamnosus strain GG sortase specificity and pilus biogenesis.J Biol Chem. 2014 May 30;289(22):15764-75. doi: 10.1074/jbc.M113.542332. Epub 2014 Apr 21. J Biol Chem. 2014. PMID: 24753244 Free PMC article.
-
Protective Immunity Against Enterotoxigenic Escherichia coli by Oral Vaccination of Engineered Lactococcus lactis.Curr Microbiol. 2021 Sep;78(9):3464-3473. doi: 10.1007/s00284-021-02601-x. Epub 2021 Jul 15. Curr Microbiol. 2021. PMID: 34264362 Free PMC article.
-
Human Gut-Commensalic Lactobacillus ruminis ATCC 25644 Displays Sortase-Assembled Surface Piliation: Phenotypic Characterization of Its Fimbrial Operon through In Silico Predictive Analysis and Recombinant Expression in Lactococcus lactis.PLoS One. 2015 Dec 28;10(12):e0145718. doi: 10.1371/journal.pone.0145718. eCollection 2015. PLoS One. 2015. PMID: 26709916 Free PMC article.
References
-
- Baumann, U. 2008. Mucosal vaccination against bacterial respiratory infections. Expert Rev. Vaccines 7:1257-1276. - PubMed
-
- Bolken, T. C., C. A. Franke, K. F. Jones, R. H. Bell, R. M. Swanson, D. S. King, V. A. Fischetti, and D. E. Hruby. 2002. Analysis of factors affecting surface expression and immunogenicity of recombinant proteins expressed by gram-positive commensal vectors. Infect. Immun. 70:2487-2491. - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

