Inhibition of PTP1B restores IRS1-mediated hepatic insulin signaling in IRS2-deficient mice
- PMID: 20028942
- PMCID: PMC2828646
- DOI: 10.2337/db09-0796
Inhibition of PTP1B restores IRS1-mediated hepatic insulin signaling in IRS2-deficient mice
Abstract
Objective: Mice with complete deletion of insulin receptor substrate 2 (IRS2) develop hyperglycemia, impaired hepatic insulin signaling, and elevated gluconeogenesis, whereas mice deficient for protein tyrosine phosphatase (PTP)1B display an opposing hepatic phenotype characterized by increased sensitivity to insulin. To define the relationship between these two signaling pathways in the regulation of liver metabolism, we used genetic and pharmacological approaches to study the effects of inhibiting PTP1B on hepatic insulin signaling and expression of gluconeogenic enzymes in IRS2(-/-) mice.
Research design and methods: We analyzed glucose homeostasis and insulin signaling in liver and isolated hepatocytes from IRS2(-/-) and IRS2(-/-)/PTP1B(-/-) mice. Additionally, hepatic insulin signaling was assessed in control and IRS2(-/-) mice treated with resveratrol, an antioxidant present in red wine.
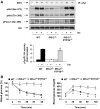
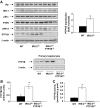
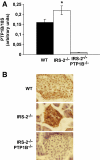
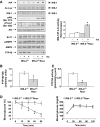
Results: In livers of hyperglycemic IRS2(-/-) mice, the expression levels of PTP1B and its association with the insulin receptor (IR) were increased. The absence of PTP1B in the double-mutant mice restored hepatic IRS1-mediated phosphatidylinositol (PI) 3-kinase/Akt/Foxo1 signaling. Moreover, resveratrol treatment of hyperglycemic IRS2(-/-) mice decreased hepatic PTP1B mRNA and inhibited PTP1B activity, thereby restoring IRS1-mediated PI 3-kinase/Akt/Foxo1 signaling and peripheral insulin sensitivity.
Conclusions: By regulating the phosphorylation state of IR, PTB1B determines sensitivity to insulin in liver and exerts a unique role in the interplay between IRS1 and IRS2 in the modulation of hepatic insulin action.
Figures



Similar articles
-
Resveratrol treatment restores peripheral insulin sensitivity in diabetic mice in a sirt1-independent manner.Mol Nutr Food Res. 2015 Aug;59(8):1431-42. doi: 10.1002/mnfr.201400933. Epub 2015 Apr 28. Mol Nutr Food Res. 2015. PMID: 25808216
-
IRS2 and PTP1B: Two opposite modulators of hepatic insulin signalling.Arch Physiol Biochem. 2011 Jul;117(3):105-15. doi: 10.3109/13813455.2011.557386. Epub 2011 Mar 14. Arch Physiol Biochem. 2011. PMID: 21401320 Review.
-
The reciprocal stability of FOXO1 and IRS2 creates a regulatory circuit that controls insulin signaling.Mol Endocrinol. 2006 Dec;20(12):3389-99. doi: 10.1210/me.2006-0092. Epub 2006 Aug 17. Mol Endocrinol. 2006. PMID: 16916938
-
Loss of protein tyrosine phosphatase 1B increases IGF-I receptor tyrosine phosphorylation but does not rescue retinal defects in IRS2-deficient mice.Invest Ophthalmol Vis Sci. 2013 Jun 19;54(6):4215-25. doi: 10.1167/iovs.12-11438. Invest Ophthalmol Vis Sci. 2013. PMID: 23702782
-
Divergent Roles of IRS (Insulin Receptor Substrate) 1 and 2 in Liver and Skeletal Muscle.Curr Med Chem. 2017;24(17):1827-1852. doi: 10.2174/0929867324666170426142826. Curr Med Chem. 2017. PMID: 28462703 Review.
Cited by
-
Leptin signaling and leptin resistance.Med Rev (2021). 2022 Aug 9;2(4):363-384. doi: 10.1515/mr-2022-0017. eCollection 2022 Aug. Med Rev (2021). 2022. PMID: 37724323 Free PMC article. Review.
-
Crosstalk between insulin and Toll-like receptor signaling pathways in the central nervous system.Mol Neurobiol. 2014 Dec;50(3):797-810. doi: 10.1007/s12035-013-8631-3. Epub 2014 Jan 25. Mol Neurobiol. 2014. PMID: 24464263 Review.
-
Protein tyrosine phosphatase 1B inhibitory activity of Indonesian herbal medicines and constituents of Cinnamomum burmannii and Zingiber aromaticum.J Nat Med. 2013 Apr;67(2):264-70. doi: 10.1007/s11418-012-0674-7. Epub 2012 May 30. J Nat Med. 2013. PMID: 22645080
-
Development of novel cell lines of diabetic dysfunction model fit for cell-based screening tests of medicinal materials.Cytotechnology. 2013 Jan;65(1):105-18. doi: 10.1007/s10616-012-9466-x. Epub 2012 Jul 10. Cytotechnology. 2013. PMID: 22777586 Free PMC article.
-
Essential role of protein-tyrosine phosphatase 1B in the modulation of insulin signaling by acetaminophen in hepatocytes.J Biol Chem. 2014 Oct 17;289(42):29406-19. doi: 10.1074/jbc.M113.539189. Epub 2014 Sep 9. J Biol Chem. 2014. PMID: 25204659 Free PMC article.
References
-
- White MF: Regulating insulin signaling and beta-cell function through IRS proteins. Can J Physiol Pharmacol 2006; 84: 725– 737 - PubMed
-
- Araki E, Lipes MA, Patti ME, Brüning JC, Haag B, 3rd, Johnson RS, Kahn CR: Alternative pathway of insulin signaling in mice with targeted disruption of the IRS1 gene. Nature 1994; 372: 186– 190 - PubMed
-
- Withers DJ, Gutierrez JS, Towery H, Burks DJ, Ren JM, Previs S, Zhang Y, Bernal D, Pons S, Shulman GI, Bonner-Weir S, White MF: Disruption of IRS2 causes type 2 diabetes in mice. Nature 1998; 391: 900– 904 - PubMed
-
- Withers DJ, Burks DJ, Towery HH, Altamuro SL, Flint CL, White MF: IRS2 coordinates IGF-I receptor-mediated β-cell development and peripheral insulin signaling. Nat Genet 1999; 23: 32– 40 - PubMed
-
- Kubota N, Tobe K, Terauchi Y, Eto K, Yamauchi T, Suzuki R, Tsubamoto Y, Komeda K, Nakano R, Miki H, Satoh S, Sekihara H, Sciacchitano S, Lesniak M, Aizawa S, Nagai R, Kimura S, Akanuma Y, Taylor SI, Kadowaki T: Disruption of insulin receptor substrate 2 causes type 2 diabetes because of liver insulin resistance and lack of compensatory β-cell hyperplasia. Diabetes 2000; 49: 1880– 1889 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

