A rhodanine agent active against non-replicating intracellular Mycobacterium avium subspecies paratuberculosis
- PMID: 20030828
- PMCID: PMC2806251
- DOI: 10.1186/1757-4749-1-25
A rhodanine agent active against non-replicating intracellular Mycobacterium avium subspecies paratuberculosis
Abstract
Background: Antibiotic therapy targeting chronic mycobacterial disease is often ineffective due to problems with the emergence of drug resistance and non-replicating persistent intracellular antibiotic resistant phenotypes. Strategies which include agents able to enhance host cell killing mechanisms could represent an alternative to conventional methods with the potential for host clearance if active against dormant phenotypes. Investigations of agents with potential activity against non-replicating mycobacteria however are restricted due to a need for assays that can assess bacterial viability without having to culture.
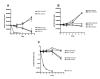
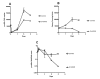
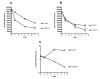
Results: This study describes the development and use of a pre16S ribosomal gene RNA/DNA ratio viability assay which is independent of the need for culture, supported by a novel thin layer accelerated mycobacterial colony forming method for determining viability and culturability of MAP in intracellular environments. We describe the use of these tools to demonstrate intracellular killing activity of a novel rhodanine agent (D157070) against the intracellular pathogen Mycobacterium avium subspecies paratuberculosis (MAP) and show that the culturability of MAP decreases relative to its viability on intracellular entry suggesting the induction of a non-culturable phenotype. We further demonstrate that D157070, although having no direct activity against the culturability of extracellular MAP, can bind to cultured MAP cells and has significant influence on the MAP transcriptome, particularly with respect of delta(L )associated genes. D157070 is shown to be taken up by bovine and human cells and able to enhance host cell killing, as measured by significant decreases in both culturability and viability of intracellular MAP.
Conclusions: This work suggests that pre16srRNA gene ratios represent a viable method for studying MAP viability. In addition, the rhodanine agent D157070 tested is non-toxic and enhances cell killing activity against both growing and latent MAP phenotypes.
Figures
Similar articles
-
Increased viability but decreased culturability of Mycobacterium avium subsp. paratuberculosis in macrophages from inflammatory bowel disease patients under Infliximab treatment.Med Microbiol Immunol. 2015 Dec;204(6):647-56. doi: 10.1007/s00430-015-0393-2. Epub 2015 Feb 22. Med Microbiol Immunol. 2015. PMID: 25702170
-
Bovine Neutrophils Release Extracellular Traps and Cooperate With Macrophages in Mycobacterium avium subsp. paratuberculosis clearance In Vitro.Front Immunol. 2021 Mar 17;12:645304. doi: 10.3389/fimmu.2021.645304. eCollection 2021. Front Immunol. 2021. PMID: 33815401 Free PMC article.
-
A 16 kb naturally occurring genomic deletion including mce and PPE genes in Mycobacterium avium subspecies paratuberculosis isolates from goats with Johne's disease.Vet Microbiol. 2012 Sep 14;159(1-2):60-8. doi: 10.1016/j.vetmic.2012.03.010. Epub 2012 Mar 13. Vet Microbiol. 2012. PMID: 22472702
-
Baseballs, tennis balls, livestock farm manure, the IDH1 mutation, endothelial cell proliferation and hypoxic pseudopalisading (granulomatous) necrosis: Mycobacterium avium subspecies paratuberculosis and the epidemiology, cellular metabolism and histology of diffuse gliomas, including glioblastoma.Open Vet J. 2019 Apr;9(1):5-12. doi: 10.4314/ovj.v9i1.2. Epub 2019 Jan 22. Open Vet J. 2019. PMID: 31086759 Free PMC article. Review.
-
Mammalian cell entry operons; novel and major subset candidates for diagnostics with special reference to Mycobacterium avium subspecies paratuberculosis infection.Vet Q. 2019 Dec;39(1):65-75. doi: 10.1080/01652176.2019.1641764. Vet Q. 2019. PMID: 31282842 Free PMC article. Review.
Cited by
-
Identification of Novel Seroreactive Antigens in Johne's Disease Cattle by Using the Mycobacterium tuberculosis Protein Array.Clin Vaccine Immunol. 2017 Jul 5;24(7):e00081-17. doi: 10.1128/CVI.00081-17. Print 2017 Jul. Clin Vaccine Immunol. 2017. PMID: 28515134 Free PMC article.
-
Phenotypic and transcriptomic response of auxotrophic Mycobacterium avium subsp. paratuberculosis leuD mutant under environmental stress.PLoS One. 2012;7(6):e37884. doi: 10.1371/journal.pone.0037884. Epub 2012 Jun 4. PLoS One. 2012. PMID: 22675497 Free PMC article.
-
Increased viability but decreased culturability of Mycobacterium avium subsp. paratuberculosis in macrophages from inflammatory bowel disease patients under Infliximab treatment.Med Microbiol Immunol. 2015 Dec;204(6):647-56. doi: 10.1007/s00430-015-0393-2. Epub 2015 Feb 22. Med Microbiol Immunol. 2015. PMID: 25702170
-
A phase 1b clinical trial to determine the safety, tolerability and immunogenicity of simian adenovirus and poxvirus vectored vaccines against a Mycobacterium avium complex subspecies in patients with active Crohn's disease.EBioMedicine. 2025 Mar;113:105570. doi: 10.1016/j.ebiom.2025.105570. Epub 2025 Feb 7. EBioMedicine. 2025. PMID: 39922068 Free PMC article. Clinical Trial.
References
-
- Singh AV, Singh SV, Makharia GK, Singh PK, Sohal JS. Presence and characterization of Mycobacterium avium subspecies paratuberculosis from clinical and suspected cases of Crohn's disease and in the healthy human population in India. Int J Infect Dis. 2008;12:190–197. doi: 10.1016/j.ijid.2007.06.008. - DOI - PubMed
-
- Deb C, Lee CM, Dubey VS, Daniel J, Abomoelak B, Sirakova TD, Pawar S, Rogers L, Kolattukudy PE. A novel in vitro multiple-stress dormancy model for Mycobacterium tuberculosis generates a lipid-loaded, drug-tolerant, dormant pathogen. PLoS ONE. 2009;4:e6077. doi: 10.1371/journal.pone.0006077. - DOI - PMC - PubMed
-
- Mura M, Bull TJ, Evans H, Sidi-Boumedine K, McMinn L, Rhodes G, Pickup R, Hermon-Taylor J. Replication and Long-Term Persistence of Bovine and Human Strains of Mycobacterium avium subsp. paratuberculosis within Acanthamoeba polyphaga. Appl Environ Microbiol. 2006;72:854–859. doi: 10.1128/AEM.72.1.854-859.2006. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

