Cyclosporine for ocular inflammatory diseases
- PMID: 20031223
- PMCID: PMC2830390
- DOI: 10.1016/j.ophtha.2009.08.010
Cyclosporine for ocular inflammatory diseases
Abstract
Purpose: To evaluate the clinical outcomes of cyclosporine treatment for noninfectious ocular inflammation.
Design: Retrospective cohort study.
Participants: A total of 373 patients with noninfectious ocular inflammation managed at 4 tertiary ocular inflammation clinics in the United States observed to use cyclosporine as a single noncorticosteroid immunosuppressive agent to their treatment regimen, between 1979 and 2007 inclusive.
Methods: Participants were identified from the Systemic Immunosuppressive Therapy for Eye Diseases Cohort Study. Demographic and clinical characteristics, including dosage of cyclosporine and main outcome measures, were obtained for every eye of every patient at every visit via medical record review by trained expert reviewers.
Main outcome measures: Control of inflammation, sustained control after reducing corticosteroid dosages, and discontinuation of therapy because of toxicity.
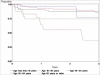
Results: Of the 373 patients (681 eyes) initiating cyclosporine monotherapy, 33.4% by 6 months and 51.9% by 1 year gained sustained, complete control of inflammation over at least 2 visits spanning at least 28 days. Approximately 25% more improved to a level of slight inflammatory activity by each of these time points. Corticosteroid-sparing success (completely controlled inflammation for at least 28 days with prednisone < or = 10 mg/day) was achieved by 22.1% by 6 months and 36.1% within 1 year. Toxicity led to discontinuation of therapy within 1 year by 10.7% of the population. Patients aged more than 55 years were more than 3-fold more likely to discontinue therapy because of toxicity than patients aged 18 to 39 years. Doses of 151 to 250 mg/day tended to be more successful than lower doses and were not associated with a higher discontinuation for toxicity rate; higher doses did not seem to offer a therapeutic advantage.
Conclusions: Cyclosporine, with corticosteroid therapy as indicated, was modestly effective for controlling ocular inflammation. Our data support a preference for cyclosporine adult dosing between 151 and 250 mg/day. Although cyclosporine was tolerated by the majority of patients, toxicity was more frequent with increasing age; alternative agents may be preferred for patients aged more than 55 years.
Copyright 2010 American Academy of Ophthalmology. Published by Elsevier Inc. All rights reserved.
Figures
Similar articles
-
Methotrexate for ocular inflammatory diseases.Ophthalmology. 2009 Nov;116(11):2188-98.e1. doi: 10.1016/j.ophtha.2009.04.020. Epub 2009 Sep 12. Ophthalmology. 2009. PMID: 19748676 Free PMC article.
-
Cyclophosphamide for ocular inflammatory diseases.Ophthalmology. 2010 Feb;117(2):356-65. doi: 10.1016/j.ophtha.2009.06.060. Epub 2009 Dec 6. Ophthalmology. 2010. PMID: 19969366 Free PMC article.
-
Mycophenolate mofetil for ocular inflammation.Am J Ophthalmol. 2010 Mar;149(3):423-32.e1-2. doi: 10.1016/j.ajo.2009.09.026. Epub 2009 Dec 30. Am J Ophthalmol. 2010. PMID: 20042178 Free PMC article.
-
Systemic therapy with conventional and novel immunomodulatory agents for ocular inflammatory disease.Surv Ophthalmol. 2011 Nov-Dec;56(6):474-510. doi: 10.1016/j.survophthal.2011.05.003. Surv Ophthalmol. 2011. PMID: 22117884 Review.
-
Treatment of chronic non-infectious uveitis and scleritis.Swiss Med Wkly. 2019 Mar 10;149:w20025. doi: 10.4414/smw.2019.20025. eCollection 2019 Feb 25. Swiss Med Wkly. 2019. PMID: 30852832
Cited by
-
Systemic Cyclosporine Therapy for Scleritis: A Proposal of a Novel System to Assess the Activity of Scleritis.Case Rep Ophthalmol. 2015 May 5;6(2):149-57. doi: 10.1159/000430490. eCollection 2015 May-Aug. Case Rep Ophthalmol. 2015. PMID: 26078748 Free PMC article.
-
Clinical outcomes of cyclosporine treatment for noninfectious uveitis.Korean J Ophthalmol. 2012 Feb;26(1):21-5. doi: 10.3341/kjo.2012.26.1.21. Epub 2012 Jan 14. Korean J Ophthalmol. 2012. PMID: 22323881 Free PMC article.
-
Corticosteroid-sparing therapy: practice patterns among uveitis specialists.J Ophthalmic Inflamm Infect. 2012 Mar;2(1):21-8. doi: 10.1007/s12348-011-0047-5. Epub 2011 Nov 6. J Ophthalmic Inflamm Infect. 2012. PMID: 22057810 Free PMC article.
-
Management of chronic ocular sarcoidosis: challenges and solutions.Clin Ophthalmol. 2018 Mar 19;12:519-532. doi: 10.2147/OPTH.S128949. eCollection 2018. Clin Ophthalmol. 2018. PMID: 29593377 Free PMC article. Review.
-
Remission of Intermediate Uveitis: Incidence and Predictive Factors.Am J Ophthalmol. 2016 Apr;164:110-7.e2. doi: 10.1016/j.ajo.2015.12.034. Epub 2016 Jan 6. Am J Ophthalmol. 2016. PMID: 26772874 Free PMC article.
References
-
- Jabs DA, Rosenbaum JT, Foster CS, et al. Guidelines for the use of immunosuppressive drugs in patients with ocular inflammatory disorders: recommendations of an expert panel. Am J Ophthalmol. 2000;130:492–513. - PubMed
-
- Okada AA. Immunomodulatory therapy for ocular inflammatory disease: a basic manual and review of the literature. Ocul Immunol Inflamm. 2005;13:335–51. - PubMed
-
-
Brunton LL, Parker KL, Murri N, Blumenthal DK, editors. Goodman & Gilman's The Pharmacological Basis of Therapeutics. 11th ed. 2006. pp. xx–xx. ed-in-chief. chapter author(s). Chapter title. AQ: provide chapter author(s), chapter title, and specific inclusive pagination
-
-
- Stepkowski SM. Molecular targets for existing and novel immunosuppressive drugs. Expert Rev Mol Med. 2000;2:1–23. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources