Molecular iodine-mediated cyclization of tethered heteroatom-containing alkenyl or alkynyl systems
- PMID: 20032861
- PMCID: PMC6255281
- DOI: 10.3390/molecules14124814
Molecular iodine-mediated cyclization of tethered heteroatom-containing alkenyl or alkynyl systems
Abstract
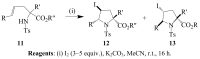
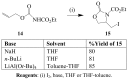
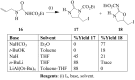
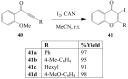
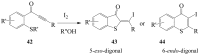
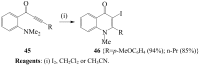
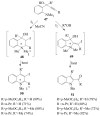
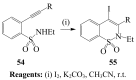
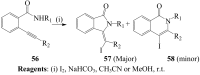
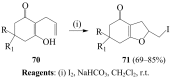
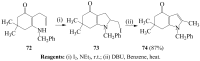
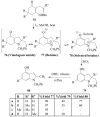
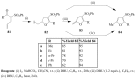
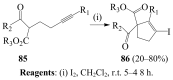
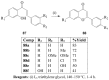
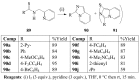
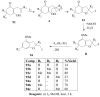
Molecular iodine has established itself as a readily available and easy-to-handle electrophilic and oxidizing reagent used in various organic transformations. In this review attention is focused on the use of molecular iodine in promoting cyclization (iodocyclization and cyclodehydroiodination) of tethered heteroatom-containing alkenyl or alkynyl systems.
Figures
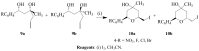
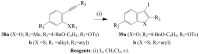
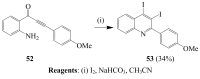
Similar articles
-
Iodine-mediated solvent-controlled selective electrophilic cyclization and oxidative esterification of o-alkynyl aldehydes: an easy access to pyranoquinolines, pyranoquinolinones, and isocumarins.J Org Chem. 2010 Nov 19;75(22):7691-703. doi: 10.1021/jo101526b. Epub 2010 Oct 29. J Org Chem. 2010. PMID: 21033701
-
A simple and mild synthesis of 1H-isochromenes and (Z)-1-alkylidene-1,3-dihydroisobenzofurans by the iodocyclization of 2-(1-alkynyl)benzylic alcohols.J Org Chem. 2010 Feb 5;75(3):897-901. doi: 10.1021/jo902333y. J Org Chem. 2010. PMID: 20043652 Free PMC article.
-
Synthesis of dibenzocyclohepten-5-ones by electrophilic iodocyclization of 1-([1,1'-biphenyl]-2-yl)alkynones.J Org Chem. 2014 Apr 18;79(8):3452-64. doi: 10.1021/jo5001803. Epub 2014 Mar 31. J Org Chem. 2014. PMID: 24641681
-
[Novel iodocyclization method based on the controlling of oxidative aromatization].Yakugaku Zasshi. 2011;131(9):1323-7. doi: 10.1248/yakushi.131.1323. Yakugaku Zasshi. 2011. PMID: 21881306 Review. Japanese.
-
Iodine-mediated synthesis of heterocycles via electrophilic cyclization of alkynes.Org Biomol Chem. 2016 Aug 10;14(32):7639-53. doi: 10.1039/c6ob01054g. Org Biomol Chem. 2016. PMID: 27383580 Review.
Cited by
-
Recent advances in the synthesis of thiophene derivatives by cyclization of functionalized alkynes.Molecules. 2014 Sep 29;19(10):15687-719. doi: 10.3390/molecules191015687. Molecules. 2014. PMID: 25268722 Free PMC article. Review.
-
Molecular iodine--an expedient reagent for oxidative aromatization reactions of alpha,beta-unsaturated cyclic compounds.Molecules. 2009 Dec 16;14(12):5308-22. doi: 10.3390/molecules14125308. Molecules. 2009. PMID: 20032894 Free PMC article. Review.
-
Recent progress in transition-metal-free functionalization of allenamides.RSC Adv. 2020 Oct 6;10(60):36818-36827. doi: 10.1039/d0ra07119f. eCollection 2020 Oct 1. RSC Adv. 2020. PMID: 35517974 Free PMC article. Review.
-
Preparation and Utility of N-Alkynyl Azoles in Synthesis.Molecules. 2019 Jan 24;24(3):422. doi: 10.3390/molecules24030422. Molecules. 2019. PMID: 30682796 Free PMC article. Review.
-
Iodine-catalyzed efficient synthesis of xanthene/thioxanthene-indole derivatives under mild conditions.RSC Adv. 2020 Jul 2;10(42):25165-25169. doi: 10.1039/d0ra05217e. eCollection 2020 Jun 29. RSC Adv. 2020. PMID: 35517437 Free PMC article.
References
-
- El-Tach G.M.M., Evans A.B., Knight D.W., Jones S. Practical alternatives for the synthesis of β-iodofurans by 5-endo-dig cyclizations of 3-alkyne-1,2-diols. Tetrahedron Lett. 2001;42:5945–5948.
-
- Bew S.P., Knight D.W. A brief synthesis of β-iodofurans. J. Chem. Soc. Chem. Commun. 1996:1007–1008. doi: 10.1039/CC9960001007. - DOI
-
- Knight D.W., Redfern A.L., Gilmore J. The synthesis of 5-substituted proline derivatives and of 5-substituted pyrrole-2-carboxylates by 5-endo cyclisations featuring a method for effectively favouring these with respect to 5-exo cyclisations. J. Chem. Soc. Perkin Trans. I. 2001:2874–2883. doi: 10.1039/b106739g. - DOI
-
- Knight D.W., Redfern A.L., Gilmore J. An approach to 2,3-dihydropyrroles and β-iodopyrroles based on 5-endo-dig cyclizations. J. Chem. Soc. Perkin Trans. I. 2002:622–628. doi: 10.1039/b110751h. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

